Hi, Benzaldehyde is easily oxidized to benzoic acid which can impede its desired reaction and hence it is always recommended to use freshly distilled benzaldehyde..
Just so, does benzaldehyde oxidize?
Benzaldehyde can be oxidized to benzoic acid, which is a common impurity in laboratory samples. Since the boiling point of benzoic acid is much higher than that of benzaldehyde, it may be purified by distillation. Benzyl alcohol can be formed from benzaldehyde by means of hydrogenation.
Secondly, what is benzaldehyde soluble in? Water
Likewise, people ask, can benzaldehyde be reduced?
Hii,,,I think you can use clemmensen reduction,. that is aldehyde and ketone is treated with zn-hg/conc HCl then it gives alkane. lialh4 will also reduce aldehyde but in alcohol not alkane so answer is no.
Does benzaldehyde react with Fehling's?
Aldehydes that lack alpha hydrogens, such as benzaldehyde or pivalaldehyde (2,2-dimethylpropanal) cannot form an enolate and thus do not give a positive Fehling's test result under usual conditions.
Related Question Answers
Is benzaldehyde saturated?
Benzaldehyde is a aromatic compound. It contains 3 double bonds in the ring. So it has to be unsaturated… Because, alkene s undergoes addition reaction while aromatic compounds including benzaldehyde undergoes substitution reaction.What is the formula of benzaldehyde?
C7H6O
Is benzaldehyde acidic or basic?
Benzaldehyde is neutral (and is neither acidic nor basic) so it would partition primarily into the organic layer at any pH. Therefore, the mixture of could be partitioned between ethyl acetate and a basic (high pH) aqueous layer.What is c7h6o?
From Wikipedia, the free encyclopedia. The molecular formula C7H6O may refer to: Benzaldehyde. Tropone.Does benzaldehyde give iodoform test?
For iodoform test one methyl must be there with carbonyl group. Thus acetaldehyde will give iodoform test and benzaldehyde does not. Thus Benzaldehyde do not give Fehling test as it do not have $$alpha-$$ Hydrogen.Does benzaldehyde give tollens test?
Fehling's solution test is given by aldehyde compounds containing an alpha hydrogen. Benzaldehyde does not give Fehling positive test while it gives a positive result with Tollen's reagent.Which is more polar benzaldehyde or benzoic acid?
All three of the substances are polar but benzoic acid has a hydroxide group that is free which makes it the most polar. Benzyl alcohol has a hydroxide group but it will bond with water. Finally, benzaldehyde the hydrogen can only bind with water while the oxygen can't.Is benzaldehyde soluble in NaOH?
1) Aromatic acids like benzoic acid and salicylic acid are NOT soluble in water. But they are soluble in 5% NaOH, and 5% NaHCO3. 2) aromatic carbonyl compounds like benzaldehyde and acetophenone are NOT soluble in H2O, 5%NaOH, 5% NaHCO3, 5% HCl.How do you convert benzaldehyde into benzophenone?
Step 1: Add the phenylmagnesium bromide along with the benzaldehyde, you will get the output as diphenylmethanol. Step 2: Add the chromyl chloride now along with the obtained diphenylmethanol, this will help you get the desired benzophenone.What are the uses of benzaldehyde?
Uses of benzaldehyde The most common use of benzaldehyde is to confer almond flavor to foods and scented products. 2. In industry, benzaldehyde is used as a precursor to other organic compounds, ranging from pharmaceuticals to plastic additives.What functional group is benzaldehyde?
aldehyde
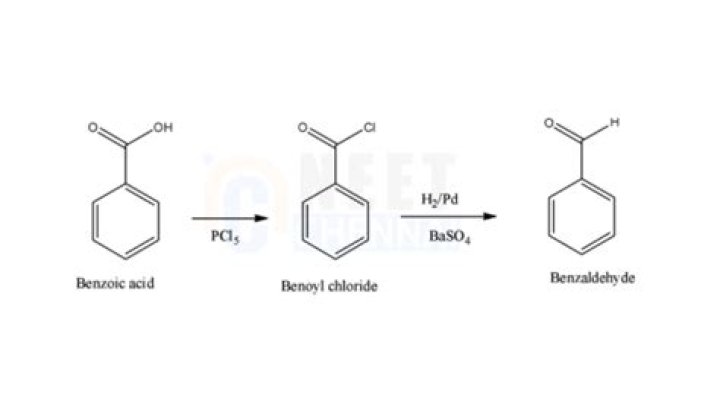
How do you purify benzaldehyde?
In accordance with the process of the present invention, purified benzaldehyde is obtained by treating the impure benzaldehyde simultaneously with water and with a metal less noble than hydrogen. The treated benzaldehyde is then distilled.How do you store benzaldehyde?
Since benzaldehyde is sensitive to air and light, it should be stored in amber bottles, which are then made air-tight by sealing them with sealing tape, like parafilm. Schlenk flasks can also be used to store this compound.Is benzaldehyde a ketone or aldehyde?
A ketone is a molecule with an R2C=O. The R groups do not have to be the same. In this reaction, two molecules of benzaldehyde (aldehyde) are condensed with one molecule of acetone (ketone). Acetone acts as a nucleophile which adds to the carbonyl carbon of benzaldehyde).What is the melting point of benzaldehyde?
-26 °C
Does benzaldehyde give silver mirror test?
Benzaldehyde is an aromatic aldehyde and it on reaction with Tollens' reagant gives silver mirror.Why is benzaldehyde not soluble in water?
benzene ring is non polar and water is a polar solvent. The lone pair on oxygen atom of benzaldehyde is completely involved in conjugation making it less available to form hydrogen bonding with water. where as lone pair on oxygen atom of acetone is readily available for hydrogen bonding and makes it more water soluble.Which is called oil of bitter almond?
Bitter Almonds come from (Prunus dulcis, var. The bitter almonds produce a small quantity of fixed oil along with an emulsion that yields glucose, cyanide, and the essential oil of bitter almonds, which is nearly pure benzaldehyde.Does benzaldehyde undergo aldol condensation?
Benzaldehyde does not undergo aldol condensation whereas acetaldehyde does. Aldol condensation involves the addition of an aldehyde (or ketonic) group of one molecule of the carbonyl compound (aldehyde or ketone) with the α-hydrogen atoms of the other. Benzaldehyde, which has no α-hydrogen atom, does not.