Can permanent hardness be removed by boiling?
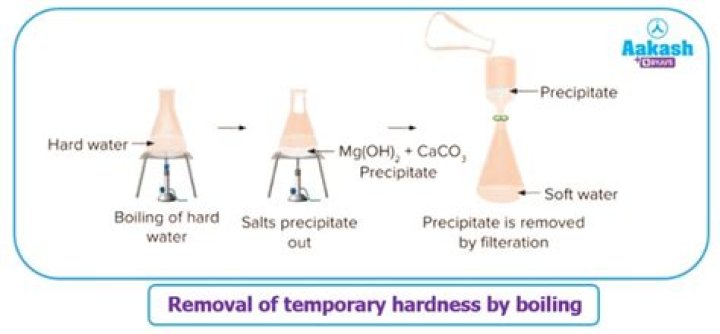
.
Also asked, can boiling remove permanent hardness of water?
Temporary hardness is caused by dissolved calcium hydrogencarbonate (which is removed by boiling). Permanent hardness is caused by dissolved calcium sulfate (which is not removed by boiling). Water can be softened using washing soda or an ion-exchange resin.
Furthermore, how can we remove permanent hardness? Permanent Hardness of Water: We can remove this hardness by treating the water with washing soda. Insoluble carbonates are formed when washing soda reacts with the sulfide and chloride salts of magnesium and calcium and thus hard water is converted to soft water.
Similarly one may ask, which type of hardness can be removed by boiling?
As you can se boiling the water causes the precipitation of solid calcium carbonate or solid magnesium carbonate. This removes the calcium ions or magnesium ions from the water, and so removes the hardness. Therefore, hardness due to hydrogencarbonates is said to be temporary.
What causes permanent hardness?
Permanent hardness in water is hardness due to the presence of the chlorides, nitrates and sulphates of calcium and magnesium, which will not be precipitated by boiling. The lime scale can build up on the inside of the pipe restricting the flow of water or causing a blockage.
Related Question AnswersHow do you remove hardness from water?
Softening is the process of removing the dissolved calcium and magnesium salts that cause hardness in water. It is achieved either by adding chemicals that form insoluble precipitates or by ion exchange. Chemicals used for softening include calcium hydroxide (slaked lime) and sodium…What are the two main causes of hardness in water?
The two main cations that cause water hardness are calcium (Ca2+) and magnesium (Mg2+). Calcium is dissolved in water as it passes over and through limestone deposits. Magnesium is dissolved as water passes over and through dolomite and other magnesium bearing formations.Can distillation remove permanent hardness of water?
Mechanical Method Mechanical water softening include: A. Distillation method - this will remove all solid particles that had dissolved in the water, but it is a rather expensive method. Both temporary and permanent hardness are removed.How do you express the total hardness of water?
When hardness is expressed as 'mg/l as CaCO3', it's calculated as if all the calcium and magnesium were present only as calcium carbonate. Hard water is a mixture of calcium and magnesium, together with bicarbonate, sulphate, chloride, etc. Ca – is calcium, C carbon and O oxygen.What are the advantage of hard water?
Advantages of hard water It is more difficult to form a lather with soap. Some people prefer the taste. Scum may form in a reaction with soap, wasting the soap. Calcium ions in the water are good for children's teeth and bones.How does sodium carbonate remove water hardness?
Sodium carbonate, Na 2CO 3, is also known as washing soda. It can soften water that has temporary hardness and it can soften water that has permanent hardness. The calcium ions come from the hard water and the carbonate ions from the washing soda.What is the unit of hardness?
The unit of hardness given by the test is known as the Vickers Pyramid Number (HV) or Diamond Pyramid Hardness (DPH). The hardness number can be converted into units of pascals, but should not be confused with pressure, which uses the same units.Does hardness increase with temperature?
The hardness of the Chrome-plated surface (normalized to 0.01 in thickness) increases gradually with temperature (Fig. This behavior is explained by the progressive decrease in grain size of the chromium with increase in temperature, which produces greater homogeneity and compactness of the deposited grains.How is temporary and permanent hardness of water removed?
Sodium carbonate, N a 2 C O 3 Na_2CO_3 Na2CO3 is also known as washing soda. It can remove temporary and permanent hardness from water. It is soluble in water and adds a large amount of carbonate ions to the water. These react with dissolved calcium and magnesium ions in the water to produce insoluble precipitates.What is the method used to remove permanent hardness in water?
Borax is usually used to reduce the alkalinity of soap solution rather than to soften water. (c) Removal of Permanent Hardness by the Base-Exchange Process: Base exchange process' is a chemical method by which, softening of permanent hardness in water can be done on a large scale or for household purposes.How is temporary and permanent hardness removed?
Soda removes both temporary and permanent hardness.Some of the methods to remove hardness from waterare,
- Chemical Process of Boiling Hard Water.
- Adding Slaked Lime (Clark's Process)
- Adding Washing Soda.
- Calgon Process.
- Ion Exchange Process.
- Using Ion Exchange Resins.


