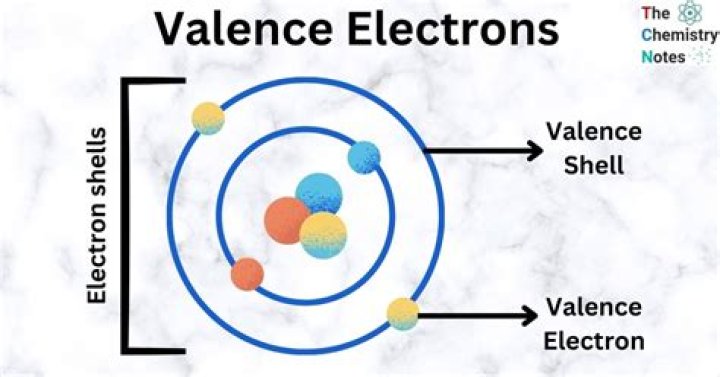
Valence Electrons. The electrons in thecloud surrounding an element's nucleus are arranged inlayers, called shells. Each shell has a specific number ofelectrons it can hold. Elements in group 1A have only onevalence electron, and each group A column to the right addsone more valence electron..
Likewise, people ask, how are electrons arranged?
The electrons in an atom move around the nucleusin regions known as electron shells. Each electronshell can only contain a certain number of electrons. Theway the electrons are arranged in an atom is called theatom's electronic structure or electronicconfiguration.
Also, why is 3rd shell 8 or 18? This is because it is the maximum capacity of the 3rdshell and it doesn't tell about the order in which theelectrons are filled. The 2,8,18,…configuration is taught till class 10th in schools. The answer toyour question lies in knowing the real configuration ofelements.
Similarly, it is asked, how do you determine valence electrons?
For neutral atoms, the number of valenceelectrons is equal to the atom's main group number. The maingroup number for an element can be found from its column on theperiodic table. For example, carbon is in group 4 and has 4valence electrons. Oxygen is in group 6 and has 6 valenceelectrons.
How is the periodic table arranged?
The periodic table of elements arranges all ofthe known chemical elements in an informative array. Elements arearranged from left to right and top to bottom in order ofincreasing atomic number. Order generally coincides with increasingatomic mass. The rows are called periods.
Related Question Answers
How many electrons are in each shell?
Each shell can contain only a fixed number ofelectrons: The first shell can hold up to twoelectrons, the second shell can hold up to eight (2 +6) electrons, the third shell can hold up to 18 (2 +6 + 10) and so on. The general formula is that the nth shellcan in principle hold up to 2(n2)electrons.What is 2n2 rule?
This rule of arrangement of electrons accordingto the shell is known 2n2 rule where n means number ofshell. For example: There is one proton in the nucleus of hydrogenatom, which moves in K shell path. It has no neutron.Are electrons in pairs?
This limits the number of electrons in the sameorbital to exactly two. The pairing of spins is often energeticallyfavorable and electron pairs therefore play a very largerole in chemistry. They can form a chemical bond between two atoms,or they can occur as a lone pair of valenceelectrons.How electrons are filled in shells?
Thus, the electron shells of an atom arepopulated from the inside out, with electrons filling up thelow-energy shells closer to the nucleus before they moveinto the higher-energy shells further out. This outermostshell is known as the valence shell, and theelectrons found in it are called valenceelectrons.How electrons are distributed in different shells?
The arrangement and distribution ofelectrons in different orbits was given by Bohr andBury. The arrangement of electrons in differentshells and sub-shells is known as the electronicconfiguration of a particular element. For instance, K shellis the first shell and it can hold up to 2(1)2 =2 electrons.Why do all unpaired electrons need to have the same spin?
Electrons are negatively charged and, as aresult, they repel each other. Electrons tend to minimizerepulsion by occupying their own orbitals, rather than sharing anorbital with another electron. For the second rule,unpaired electrons in singly occupied orbitals have thesame spins.What's an orbital diagram?
Orbital diagrams are pictorial descriptions ofthe electrons in an atom. Three rules are useful in formingorbital diagrams. According to the Auf Bau Principle, eachelectron occupies the lowest energy orbital. Orbitaldiagrams are a pictorial description of electrons in anatom.Why are paired electrons more stable?
Pairing the electrons in the same orbitalwould place them in closer proximity (hence higher energy) thanplacing them in two different orbitals where they remainunpaired. A further consequence of having unpairedelectrons is that it makes the moleculesparamagnetic.What do valence electrons do?
Valence electrons are the electronslocated at the outermost shell of an atom. Because when two atomsinteract, the electrons in the outermost shells are thefirst ones to come into contact with each other and are the onesthat determine how an atom will react in a chemicalreaction.How many valence electrons does f have?
7 valence electrons
How many valence electrons does C have?
four valence electrons
What do you mean by Valency?
In chemistry, the valence or valency of anelement is a measure of its combining power with other atoms whenit forms chemical compounds or molecules.How many valence electrons does Zr have?
4 valence electrons
How is Valency calculated?
To calculate the valency of an element(ormolecule, for that matter), there are multiple methods. Thevalency of an atom is equal to the number of electrons inthe outer shell if that number is four or less. Otherwise, thevalency is equal to eight minus the number of electrons inthe outer shell.How many valence electrons does zinc have?
two valence electrons
How many electrons can the 8th Shell hold?
The L shell only holds eight electrons.The M shell only holds eight electrons. The Mshell can actually hold up to 18 electrons asyou move to higher atomic numbers. The maximum number ofelectrons you will find in any shell is32.Why are there 8 electrons in the third shell?
The third shell is the outer valenceshell, so it has 5 valence electrons. The number ofelectrons in each shell becomes more complicated asmore electrons are added because there are moresubshells being used and because the shell start to fill outof order.How many electrons can 3p hold?
six electrons
Can there be more than 8 electrons in a shell?
Some molecules can accommodate more than 8valence electrons because they have expanded valenceshells. These molecules are from periods 3 or higherbecause starting from n=3, atoms have d-orbitals that canaccommodate for more that 9 valenceelectrons.