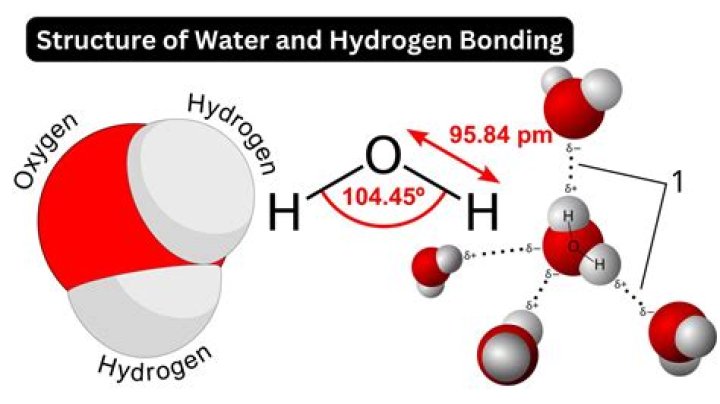
Water is an incredible small molecule when compared other molecules. It's approximate diameter is 2.75 angstroms. There is 100 picometers to 1 angstrom so this is 275 picometers or 0.275 nanometers..
Consequently, what is the size of a molecule?
Molecular size Molecules commonly used as building blocks for organic synthesis have a dimension of a few angstroms (Å) to several dozen Å, or around one billionth of a meter.
One may also ask, are all water molecules the same size? Molecules have size and shape. Atoms bond together to form molecules that have different sizes and shapes. All water molecules have the same shape because the bonds between the hydrogen atoms and the oxygen atom are more or less the same angle. Single molecules can be made up of thousands and thousands of atoms.
Also question is, how many molecules are in a drop of water?
So, next we calculate how many molecules there are in a drop of water, which we determined contains 0.002775 moles: molecules in a drop of water = (6.022 x 1023 molecules/mole) x 0.002275 moles. molecules in a drop of water = 1.67 x 1021 water molecules.
How big is a molecule in inches?
Measuring in Nanometers In addition to mass, we can also measure how big a molecule is. A water molecule is about 0.27 of a nanometer across. DNA is about 2 nanometers across and can stretch out to meters in length.
Related Question Answers
What are 3 examples of molecules?
Examples of Molecules: - Carbon dioxide - CO2
- Water - H2O.
- Oxygen we breathe into our lungs - O2
- Sugar - C12H22O11
- Glucose - C6H12O6
- Nitrous oxide - "Laughing gas" - N2O.
- Acetic acid - part of vinegar - CH3COOH. Related Links: Examples. Science Examples.
How heavy is a molecule?
Since atomic weights are average values, molecular weights are also average values. On the average, a molecule of ordinary water weighs 18.015 amu. Both hydrogen and oxygen are made up of several isotopes.How many different types of molecules are there?
There are three types of molecules which are the element molecule, the compound molecule & the mixture.How big is a protein molecule?
The average size of a protein increases from Archaea to Bacteria to Eukaryote (283, 311, 438 residues and 31, 34, 49 kDa respecitvely) due to a bigger number of protein domains constituting proteins in higher organisms. For instance, yeast proteins are on average 466 amino acids long and 53 kDa in mass.Do molecules change size?
Molecules are held together by bonds, so increasing a molecules size means you have to stretch these bonds. Matter is made up of atoms and molecules that are constantly moving. When heat is added to a substance, the molecules and atoms vibrate faster. As atoms vibrate faster, the space between atoms increases.How small is a molecule?
Small molecule. Within the fields of molecular biology and pharmacology, a small molecule is a low molecular weight (< 900 daltons) organic compound that may regulate a biological process, with a size on the order of 1 nm. Many drugs are small molecules.How many atoms are in a molecule?
2 atoms
What is the smallest to largest molecule?
Some of the largest molecules are macromolecules or supermolecules. The smallest molecule is the diatomic hydrogen (H2), with a bond length of 0.74 Å. Effective molecular radius is the size a molecule displays in solution.How many drops of water are in a gallon?
75,708.24 drops
How many atoms are in the human body?
In summary, for a typical human of 70 kg, there are almost 7*1027 atoms (that's a 7 followed by 27 zeros!) Another way of saying this is "seven billion billion billion." Of this, almost 2/3 is hydrogen, 1/4 is oxygen, and about 1/10 is carbon. These three atoms add up to 99% of the total!How many molecules are in a gram of water?
or 1 g of water contains (6.022 x 10^23)/ 18 water molecules and the number comes out to be 3.3455 x 10^22. So the number of water molecules present in 1 g of water is 3.3455 x 10^22.How many molecules are in a gram?
A mass in grams numerically equal to the molecular weight contains one mole of molecules, which is known to be 6.02 x 10^23 (Avogadro's number). So if you have x grams of a substance, and the molecular weight is y, then the number of moles n = x/y and the number of molecules = n multiplied by Avogadro's number.How many atoms are in h2o?
For H2O, there is one atom of oxygen and two atoms of hydrogen. A molecule can be made of only one type of atom. In its stable molecular form, oxygen exists as two atoms and is written O2. to distinguish it from an atom of oxygen O, or ozone, a molecule of three oxygen atoms, O3.How many atoms are in a water molecule?
three atoms
What is a mole of water?
Mole (unit) Thus, for example, one mole of water contains 6.02214076×1023 molecules, whose total mass is about 18.015 grams – and the mean mass of one molecule of water is about 18.015 daltons. The mole is widely used in chemistry as a convenient way to express amounts of reactants and products of chemical reactions.How many molecules are in a mole?
Avogadro's number is a very important relationship to remember: 1 mole = 6.022×1023 6.022 × 10 23 atoms, molecules, protons, etc. To convert from moles to atoms, multiply the molar amount by Avogadro's number. To convert from atoms to moles, divide the atom amount by Avogadro's number (or multiply by its reciprocal).How many water molecules are there in a drop of volume 0.05 ml?
Explanation: That means it contains 0.01 moles of water molecules, or 6 x molecules. 1 drop is roughly equal to 1/20 ml = 0.05 ml.What type of bond is water?
Water is a polar molecule A water molecule is formed when two atoms of hydrogen bond covalently with an atom of oxygen. In a covalent bond electrons are shared between atoms. In water the sharing is not equal. The oxygen atom attracts the electrons more strongly than the hydrogen.How long do water molecules last?
A typical water molecule will stick around in an ocean for, on average, a few thousand years. In rivers, a water molecule won't dawdle as long — just a couple weeks to several months. But a water molecule hunkered down in groundwater might be around for 10,000 years.