How do you determine if a substance is acidic or basic?
.
Simply so, how do you determine if a solution is acidic or basic?
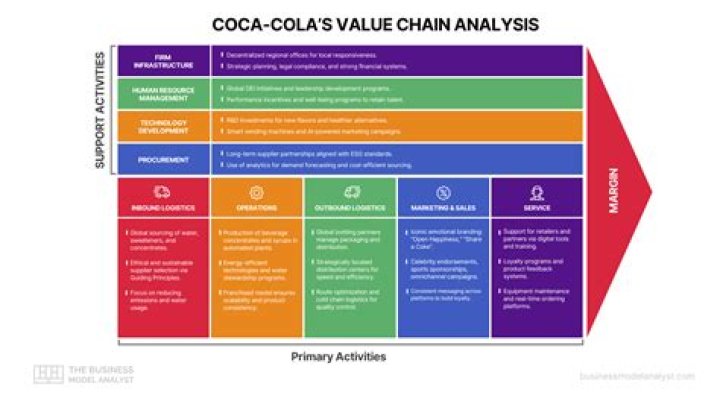
A solution with a pH of 7 is classifiedasneutral. If the pH is lower than 7, the solutionisacidic. When pH is higher than 7, the solutionisbasic. These numbers describe the concentration ofhydrogenions in the solution and increase on a negativelogarithmicscale.
how do you know if a salt is acidic or basic? The pH of a solution reflects the concentration ofH3O+ions. A pH less than 7 indicates an acidic solution, apHgreater than 7 indicates a basic solution, and a pH of7indicates a neutral solution. Acidic saltsmakeacidic solutions and basic salts makebasicsolutions.
Regarding this, how do you determine if an acid is strong or weak?
If an acid is not listed here, it is aweakacid. It may be 1% ionized or 99% ionized, but it isstillclassified as a weak acid. Any acid thatdissociates100% into ions is called a strong acid. Ifit doesnot dissociate 100%, it is a weak acid.
Is LiOH an acid or base?
No, LiOH is a strong base, like theotheralkali bases. It is a bit weaker than bases likeNaOHthough, because it's enthalpy of formation is different.Itsconjugate acid, Li+, can act as a weakLewisacid.
Related Question AnswersIs Hcooh an acid or base?
The protonated form HCOOH is the acidandthe deprotonated form HCOO- is theconjugatebase. That is formic acid (HCOOH) andtheformate ion (HCOO-) are a conjugate pair. In thesamereaction, the water is the base (deprotonated)whileH3O+ (the protonated form) is theconjugateacid.Is ch3cooh an acid or base?
So a strong acid (e.g. HCl) iscompletelydissociated in water and its conjugate base(Cl–) is avery, very weak base and shows no tendanceto accept aproton. A weak acid (e.g. CH3COOH) is inequilibriumwith its ions in water and its conjugate (CH3COO–,a weakbase) is also in equilibrium in water.Is NaOH an acid or base?
NaOH, or sodium hydroxide, is a compound.Acompound is classified as either an acid, base,orsalt. All bases contain OH- (hydroxide) ions, whileallacids contain H+ (hydrogen) ions. A salt is a compoundthatis formed when a base and an acid arecombinedbecause they neutralize each other.Is Na2CO3 an acid or base?
Na2CO3 is neither an acid norabase. It is a salt. A salt in chemistry is formed bythereaction of an acid and a base. Now depending onthestrength of the respective acid and base fromwhichthe salt is derived, it may be categorised as anacidic,basic or a neutral salt.Is CaCl2 an acid or base?
Can CaCl2 be classified as an acid or asabase? CaCl2 is a salt of strong acid (HCl) andstrongbase Ca(OH)2. Thus when dissolved in water itshowsfollowing reaction. As solution contains equal number of H+and OH-ions,it is neutral in nature.What is a basic solution?
A basic solution is an aqueoussolutioncontaining more OH-ions thanH+ions. In otherwords, it is an aqueous solutionwith a pH greater than7.Is h3po4 a strong or weak acid?
Phosphorus is not electronegative enough. Along withthehigher electronegativity of N relative to P, the extra oxygenmakesHNO3 a strong acid, while the lack thereof makesH3PO4weak. After H3PO4 does lose a proton, it formsH2PO4-,which lacks sufficient resonance stabilization.Is LiOH strong or weak?
Classifying Electrolytes| Strong Electrolytes | strong acids | HCl, HBr, HI, HNO3,HClO3,HClO4, andH2SO4 |
|---|---|---|
| strong bases | NaOH, KOH, LiOH, Ba(OH)2, andCa(OH)2 | |
| salts | NaCl, KBr, MgCl2, and many, many more | |
| Weak Electrolytes | ||
| weak acids | HF, HC2H3O2 (aceticacid),H2CO3 (carbonicacid),H3PO4 (phosphoric acid), and manymore |
Is HClO4 an acid or base?
An example is perchloric acid,HCLO4 .Otherexamples of strong acids are H2SO4,HI,HCl and HNO3. Astrongbase completely ionizes in aqueous solution to giveOH- and acation . Sodium hydroxide is an example ofstrongbase.Is HCl a strong acid?
A strong acid is an acid whichiscompletely ionized in an aqueous solution. Hydrogenchloride(HCl) ionizes completely into hydrogen ions andchlorideions in water. Weak acids, like strong acids,ionizeto yield the H+ ion and a conjugate base.BecauseHCl is a strong acid, its conjugatebase(Cl−) is extremely weak.What are some examples of a weak acid?
A weak acid is an acid that doesn'tproducemany hydrogen ions when in aqueous solution. Weakacids haverelatively low pH values and are used toneutralize strongbases. Examples of weak acidsinclude: aceticacid (vinegar), lactic acid, citricacid,and phosphoric acid.What are the strong bases?
Here is a list of the most common strong bases.- LiOH - lithium hydroxide.
- NaOH - sodium hydroxide.
- KOH - potassium hydroxide.
- RbOH - rubidium hydroxide.
- CsOH - cesium hydroxide.
- *Ca(OH)2 - calcium hydroxide.
- *Sr(OH)2 - strontium hydroxide.
- *Ba(OH)2 - barium hydroxide.