The stable nucleus has approximately a constantdensity and therefore the nuclear radius R can beapproximated by the following formula, where A = Atomic mass number(the number of protons Z, plus the number of neutrons N) andr0 = 1.25 fm = 1.25 ×10−15 m..
Also to know is, how do you find the radius of the nucleus of an atom?
Most nuclei are approximately spherical. Theaverage radius of a nucleus with A nucleons is R =R0A1/3, whereR0 = 1.2*10-15 m. The volume ofthe nucleus is directly proportional to the total number ofnucleons. This suggests that all nuclei have nearly the samedensity.
what is the diameter of nucleus? The size (diameter) of the nucleus isbetween 1.6 fm (10−15 m) (for a protonin light hydrogen) to about 15 fm (for the heaviest atoms, such asuranium). These sizes are much smaller than the size of the atomitself by a factor of about 23,000 (uranium) to about 145,000(hydrogen).
One may also ask, what is the radius of a nucleus compared to an atom?
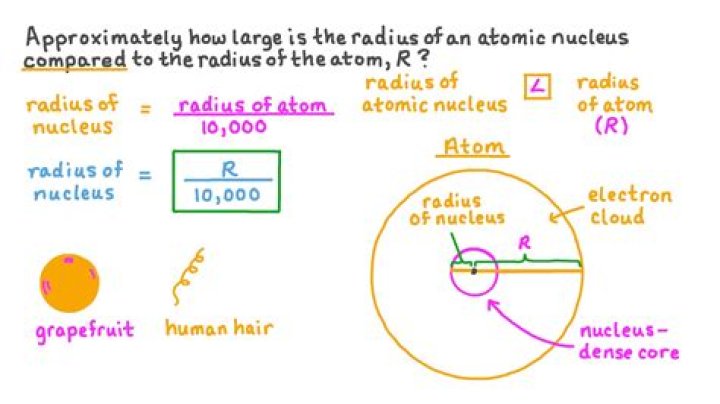
The nucleus of an atom is about10-15 m in size; this means it is about10-5 (or 1/100,000) of the size of the wholeatom. A good comparison of the nucleus to theatom is like a pea in the middle of a racetrack.(10-15 m is typical for the smallernuclei; larger ones go up to about 10 timesthat.)
What is the radius of a neutron?
The neutron has a mean square radius ofabout 0.8×10−15 m, or 0.8 fm, andit is a spin-½ fermion.
Related Question Answers
What is the radius of an atom?
The atomic radius of a chemical element is ameasure of the size of its atoms, usually the mean ortypical distance from the center of the nucleus to the boundary ofthe surrounding shells of electrons. Electrons do not have definiteorbits, or sharply defined ranges.Who discovered nucleus?
Ernest Rutherford
What is the shape of nucleus?
The shape of an atomic nucleus reflectsthe shell structure of the protons and neutrons of which it isformed. If the shells are completely filled, we speak of a "magic"nucleus, which is spherical in shape. Mostnuclei, however, tend to be deformed because their shellsare only partially filled.Where is the nucleus found?
The nucleus is an organelle that contains thegenetic information for that organism. In an animal cell, thenucleus is located in the central region of the cell.In a plant cell, the nucleus is located more on theperiphery due to the large water-filled vacuole in the center ofthe cell.Is the nucleus positively charged?
An atom consists of a positively charged nucleus,surrounded by one or more negatively charged particlescalled electrons. The positive charges equal the negativecharges, so the atom has no overall charge; it iselectrically neutral. The nucleus of an atom containsprotons and neutrons.How big is a atom?
An atom is one of the basic units of matter.Everything around us is made up of atoms. An atom isa million times smaller than the thickest human hair. The diameterof an atom ranges from about 0.1 to 0.5 nanometers (1× 10−10 m to 5 ×10−10 m).What is the nuclear radius?
The volume of a nucleus is therefore approximatelyproportional to the number of particles in the nucleus, protons andneutrons, collectively known as nucleons. For Na (11 protons, 12neutrons) the nuclear radius is 3.7 x10-15 m and the atomic radius is 1.86x 10-10m. The nucleus is 40,000 times smallerthan the atom.Which is the smallest subatomic particle?
The smallest particle is the quark, the basicbuilding block of hadrons. There are two types of hadrons: baryons(three quarks) and mesons (one quark, one antiquark). Protons andthe neutrons are stable baryons.Why is an atom neutral?
Atoms are electrically neutral becausethey have equal numbers of protons (positively charged) andelectrons (negatively charged). If an atom gains or losesone or more electrons, it becomes an ion. If it gains one or moreelectrons, it now carries a net negative charge, and is thus"anionic."How big is a quark?
While the size of protons and neutrons is of the orderof a Fermi (10−15 m), the size ofquarks is ~10−18 m. It is deemedthat quarks are composed of smaller particles –preons.What is the size of a molecule?
Molecular size Molecules commonly used as building blocks fororganic synthesis have a dimension of a few angstroms (Å) toseveral dozen Å, or around one billionth of ameter.How big is the nucleus of a cell?
Structures. The nucleus is the largest organellein animal cells. In mammalian cells, the averagediameter of the nucleus is approximately 6 micrometres(µm), which occupies about 10% of the total cellvolume.