How do you purify Benzocaine
a) Add a few mL of hot ethanol and heat the mixture on the steam bath until all the oil dissolves. b) Add water (drop wise!) to the alcohol solution until cloudiness just appears and then add a few drops of ethanol. Cool the mixture, with occasional VIGOROUS swirling, in an ice bath.
How do you synthesize benzocaine?
Synthesis of benzocaine: Into a 100 ml flask, mix 1.25 g of p-aminobenzoic acid hydrochloride, 10 ml of EtOH, and 0.5 ml H2SO4 (conc.). Heat the mixture at reflux for 2 h. After cooling the mixture, neutralize with an aqueous sodium carbonate solution (10%).
What is benzocaine made of?
Benzocaine is a chemical compound derived from para-aminobenzoic acid and ethanol. It is a white, odorless, crystalline powder. It acts on neuronal membranes and blocks nerve signals in the body. Its numbing effects are temporary.
Why do we add sodium carbonate at the end during synthesis of benzocaine?
After the solution has cooled to room temperature, add the 10% sodium carbonate solution dropwise to neutralize the cooled reaction mixture. As the pH of the solution rises carbon dioxide will be produced and evolve as gas bubbles out of the solution.Why is absolute ethanol used in the synthesis of benzocaine?
Benzocaine is soluble in Ethanol, Methanol and Diethyl Ether. But it is not very soluble in water. Having water in the solution would decrease the solubility and would create a lot of hassle later. … Using 95 % ethanol would reduce the yield of benzocaine.
Is benzocaine an acid or base?
Benzocaine is a benzoate ester having 4-aminobenzoic acid as the acid component and ethanol as the alcohol component.
Is Benzocaine a blood thinner?
Benzocaine, a local anesthetic, can cause a condition in which the amount of oxygen carried through the blood is greatly reduced. This condition, called methemoglobinemia, can be life-threatening and result in death.
How much alcohol do you put in the round bottom flask?
The distillation flask is a round-bottom flask. The liquid to be distilled should fill the distillation flask to ~50-60% of its capacity. To promote even heating of the liquid, a boiling chip or a magnetic stir bar is added before heat is applied to the distillation flask.Is benzocaine soluble in water?
Benzocaine occurs in two forms: a crystalline salt (benzocaine HCl) that is soluble in water at 0.4 g/L, and a nonwater-soluble basic form that must be dissolved in ethyl alcohol at a concentration of 0.2 g/ml before it is sufficiently soluble in water to create a functional induction dose.
How does Le Chatelier's principle influence the benzocaine synthesis?Le Chatelier’s Principle can be exploited to push the reaction toward the product side. … In this synthesis of benzocaine, by using microwave heating, we will be able to successfully complete the actual reaction of benzocaine in just a couple of minutes.
Article first time published onDoes benzocaine make you last longer?
After 2 months, the researchers found that the men using the benzocaine wipes reported a much longer IELT than men who used the placebo wipes — well over the 2-minute mark typical of premature ejaculation. The men who used the benzocaine wipes also reported: feeling less distressed by their IELT.
What color is benzocaine?
Benzocaine is a colorless trapezial crystal. Its melting point is 92℃ (88-90℃), boiling point is 183-184°C (1.87kPa). 1g of this drug is soluble in about 2500ml water, 5ml ethanol, 2ml chloroform, 4ml ether or 30-50ml almond oil and olive oil, and it is also soluble in dilute acid.
What's better benzocaine or lidocaine?
Lidocaine and benzocaine were equally efficient, and both were better than placebo in reducing pain caused by insertion of needles into the palate.
Why is ethanol a good solvent for Fischer esterification?
2. The ethanol is probably an excess reactant, so it also serves as a solvent. The most important reason for using absolute ethanol is that the Fischer esterification is an equilibrium reaction with a Keq≈4 . … The absolute alcohol contains no water, so the reaction will be able to go further to the right.
What is the difference between absolute ethanol and 95 ethanol?
Absolute ethanol is hygroscopic (it attracts water), so don’t expect it to remain ‘100%’ ethanol for long if it’s left uncapped. Denatured ethanol (either 95% or absolute) contains additives (such as methanol and isopropanol) that render it unsafe to drink, and therefore exempt from certain beverage taxes.
Why benzocaine precipitates during the neutralization?
Explain why benzocaine precipitates during the neutralization. With the addition of an acid, the amine group on the benzocaine will become protonated. The protonation creates a positive charge on the benzocaine molecule. … Thus, the benzocaine will no longer be soluble and will precipitate during neutralization.
What happens if you use too much benzocaine?
Seek emergency medical attention or call the Poison Help line at 1-800-222-1222. An overdose of benzocaine topical applied to the skin can cause life-threatening side effects such as uneven heartbeats, seizure (convulsions), coma, slowed breathing, or respiratory failure (breathing stops).
Is it illegal to have benzocaine?
7. Benzocaine and lidocaine are however legal to import and sell as bulk chemicals. They are used within the pharmaceutical industry as active substances in a number of medicinal products.
What is another name for benzocaine?
Clinical dataTrade namesAnbesol, Lanacane, Orajel, othersAHFS/Drugs.comMonographRoutes of administrationTopical, Oral
Is benzocaine the same as lidocaine?
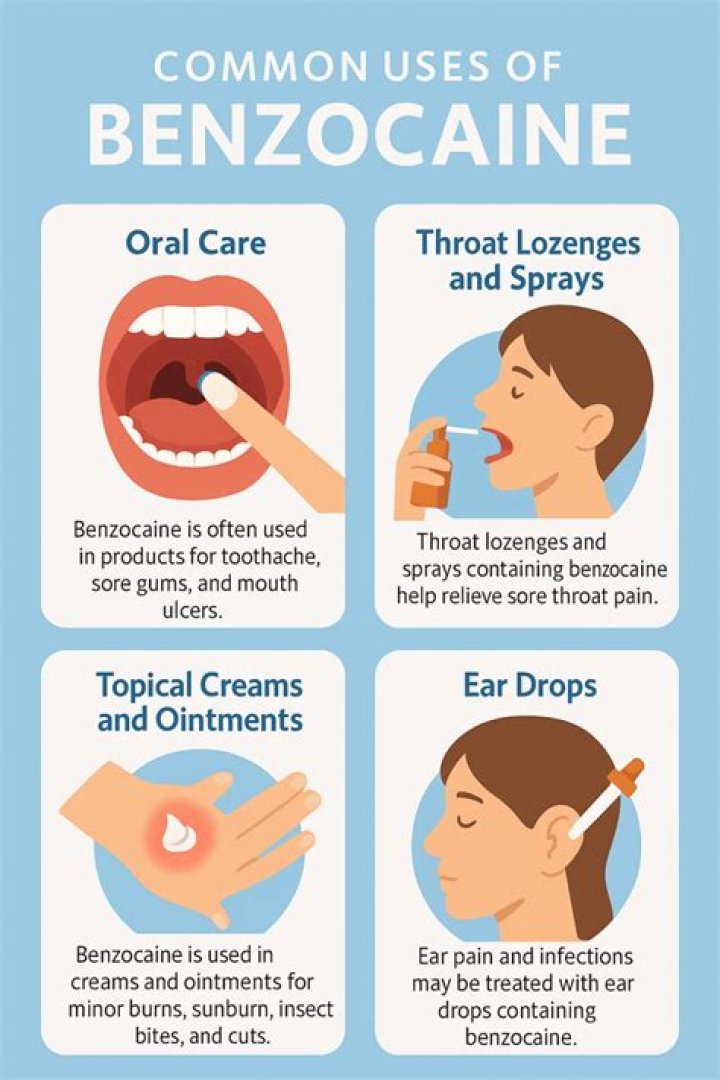
Lidocaine also comes in a systemic injection that’s used to treat cardiac rhythm problems. Benzocaine is more commonly used in throat lozenges and as a spray. Lidocaine and benzocaine are equally effective for pain relief. Both drugs have similar side effects, most commonly stinging or local skin issues.
Is benzocaine soluble in any other solvents?
Benzocaine is a surface anesthetic that reversibly inactivates voltage-dependent sodium channels. Solubility : Soluble in water (0.4 mg/ml at 20° C), methanol, chloroform (500 mg/ml), ethanol (50 mg/ml), and ether (250 mg/ml).
Is benzocaine a liquid?
Label: CVS ORAL ANESTHETIC- benzocaine liquid.
How long does benzocaine last for?
Benzocaine is a water-insoluble ester-type local anesthetic agent that is mainly useful for topical application. The onset of action is rapid (<1 minute), and the effective duration is 5 to 10 minutes.
What is benzocaine powder?
Benzocaine, sold as a powder, is an antiseptic used to numb tooth pain. That numbing is similar to the effect that cocaine has on the nose. Benzocaine can be mixed with cocaine in a 50-50 ratio without a noticeable difference in drug quality, making it the cutting agent of choice for many drug dealers.
Can benzocaine make you turn blue?
It is a rare occurrence. It’s not as if everyone using benzocaine is turning blue. Therefore, if you are less than two years old and can somehow read this, you may want to lay off on the benzocaine since your risk of methemoglobinemia is higher.
Is it illegal to carry a flask?
Generally, yes. Having a flask in your possession while driving violates open container laws. Depending on your state, you may be able to carry one as a passenger in a vehicle, but in California, this is illegal as well.
Why are 1 or 2 boiling stones added to the round bottom flask?
The alcohol vaporizes at the bottom of the round flask with no boiling chips. If boiling chips are added, then the surface area of places to boil increases, thus the mixture will boil rapidly, causing danger.
Is it safe to store alcohol in stainless steel?
Stainless steel flasks are not intended for long-term alcohol storage. There are no serious health risks associated with drinking liquor that has been stored in a stainless steel flask for a long period of time.
How do you reverse Fischer esterification?
Acid-catalyzed ester hydrolysis is literally just the reverse of Fischer esterification. It’s literally just the reverse reaction of producing an ester. You could then hydrolyze that ester back to a carboxylic acid.
How do you drive Fischer esterification to the right?
According to Le Châtelier’s Principle, we can shift the position of equilibrium to the right either by adding an excess of a reactant or by removing one of the products. If we use a large excess of one reactant (usually the cheaper one!), we can push the position of equilibrium to the side that produces more ester.
How can you increase the efficiency of Fischer esterification?
The yield of ester can be improved by increasing the concentration of one of the reactants (either the alcohol or the carboxylic acid). By Le Chatelier’s Principle an excess of one reactant will drive the reaction to the right, increasing the production of ester, and therefore increasing the yield of ester.