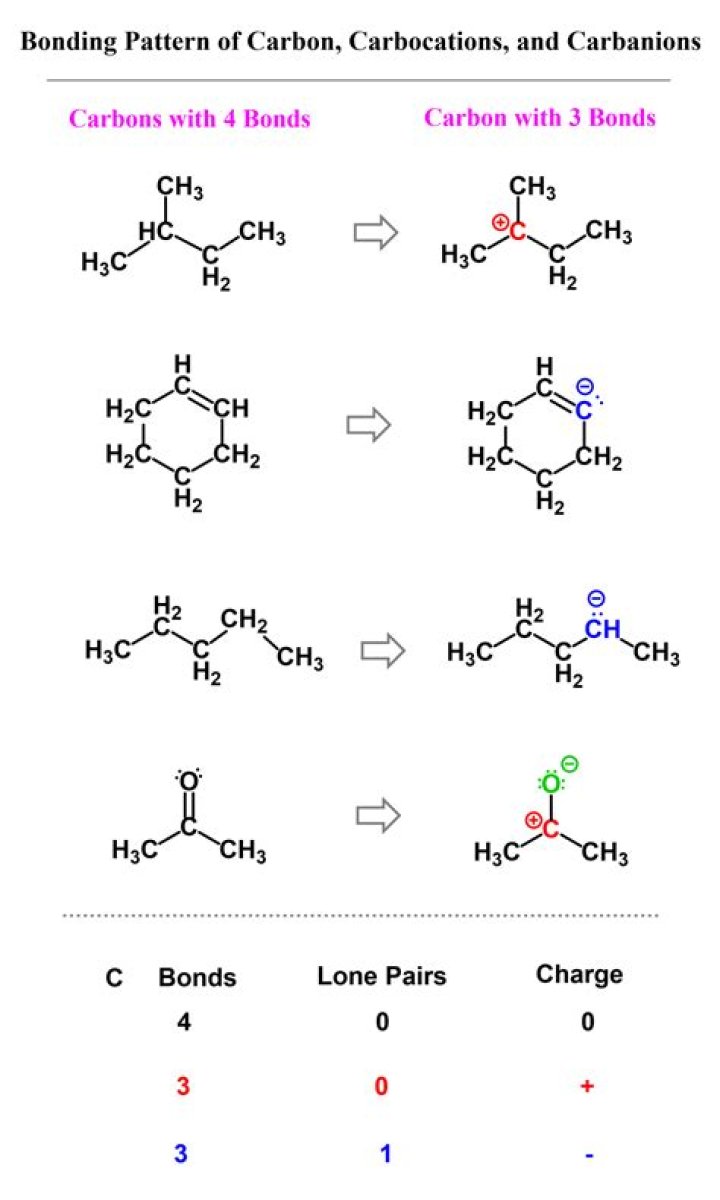
Carbon is an element capable of making bonds with other carbon atoms or atoms of other elements. Carbon has four valence electrons that can form four bonds..
Likewise, people ask, how many bonds can a carbon atom form?
The three major types of covalent bonds are single, double, and triple bonds. A carbon atom can form the following bonds: Four single bonds. One double and two single bonds.
Beside above, which kind of bond does the carbon atom form? Covalent Bonds
Furthermore, how many covalent bonds can each carbon atom form quizlet?
-In carbon compounds, carbon has 4 valence electrons and hydrogen has 1. To achieve octet, C forms 4 bonds. -VSEPR theory predicts that carbon atom with four single, covalent bonds has tetrahedral shape.
What is the maximum number of other atoms that a single carbon atom can bond to?
four bonds
Related Question Answers
Why carbon carbon Tetrabond is not possible?
Carbon never forms tetra-bond with other carbon atom. Due to repulsion between the valence electrons & nucleus of both the carbon atom.What are the 4 types of carbon bonds?
Carbon can form single bonds (sharing of 2 electrons), double bonds (sharing of 4 electrons), and/or a triple bond (sharing of 6 electrons).How many times can oxygen bond?
Oxygen has 2 missing electrons in the valence shell. Therefore it can only form 2 bonds at the most, if both are sigma bonds.What are the 5 types of chemical bonds?
Chemical bonds are forces that hold atoms together to make compounds or molecules. Chemical bonds include covalent, polar covalent, and ionic bonds. Atoms with relatively similar electronegativities share electrons between them and are connected by covalent bonds.How many bonds are in hydrogen?
The number of electrons required will be the number of bonds formed most of the time. A hydrogen atom can form 1 bond, a carbon atom can form 4 bonds, a nitrogen atom can form 3 bonds and an oxygen atom can form 2 bonds.How many times does nitrogen bond?
As known, nitrogen could form 3 bonds based on octet rule, because it has 5 valence electrons. That means it needs 3 bonds.Can two carbon atoms bond?
A carbon–carbon bond is a covalent bond between two carbon atoms. The most common form is the single bond: a bond composed of two electrons, one from each of the two atoms. Carbon atoms can also form double bonds in compounds called alkenes or triple bonds in compounds called alkynes.How many holes will the balls representing carbon have?
We will work with wooden balls to represent atoms. Hydrogens (white or yellow) have one hole, oxygens (red) have two holes, nitrogens (blue) have three holes, and carbons (grey) have four holes.What is the name of the smallest alkyne?
acetylene
How are alkanes produced?
Because alkanes contain only carbon and hydrogen, combustion produces compounds that contain only carbon, hydrogen, and/or oxygen. Like other hydrocarbons, combustion under most circumstances produces mainly carbon dioxide and water. Branched-chain alkanes are called isoparaffins.How many covalent bonds are in an alkane?
Alkanes. Alkanes, or saturated hydrocarbons, contain only single covalent bonds between carbon atoms. Each of the carbon atoms in an alkane has sp3 hybrid orbitals and is bonded to four other atoms, each of which is either carbon or hydrogen.How many double covalent bonds are in an alkene?
A double bond in chemistry is a covalent bond between two atoms involving four bonding electrons instead of the usual two. Double bonds occur most commonly between two carbon atoms, for example in alkenes.What is the general formula for a straight chain alkane?
The general formula for the straight-chain alkanes is H - (CH2)n - H. Methane (n=1) is the first member of the homologous series of the alkanes, followed by ethane (n=2) and so forth.How is an organic compound that contains only carbon and hydrogen and at least one carbon carbon triple bond classified?
The simplest organic compounds are the hydrocarbons, which contain only carbon and hydrogen. Alkanes contain only carbon–hydrogen and carbon–carbon single bonds, alkenes contain at least one carbon–carbon double bond, and alkynes contain one or more carbon–carbon triple bonds.What is the simplest straight chain alkane?
Alkanes. An alkane is a hydrocarbon in which there are only single covalent bonds. The simplest alkane is methane, with the molecular formula CH4. The carbon is the central atom and makes four single covalent bonds to hydrogen atoms.What is the physical state of the smallest alkanes at room temperature?
The alkanes can exist as gases, liquids, or solids at room temperature. The unbranched alkanes methane, ethane, propane, and butane are gases; pentane through hexadecane are liquids; the homologues larger than hexadecane are solids.Which carbon bond is the strongest?
Electronegativity and bond strength The partial charges on the fluorine and carbon are attractive, contributing to the unusual bond strength of the carbon–fluorine bond. The bond is labeled as "the strongest in organic chemistry," because fluorine forms the strongest single bond to carbon.Which bond is stronger CC or CN?
C-C with bond energy 346 kJ/mol is stronger than C-N (bond energy 305 kJ/mol). Both are strong bonds. The CN double bond and triple bond are stronger than the corresponding CC bonds, however. Ionic character or polarity in C-N single bond does not necessarily reduce the bond strength.Why does carbon bond so easily?
Because each carbon is identical, they all have four valence electrons, so they can easily bond with other carbon atoms to form long chains or rings. In fact, a carbon atom can bond with another carbon atom two or three times to make double and triple covalent bonds between two carbon atoms.