How many covalent bonds are there in c2h6?
.
Likewise, how many covalent bonds are in ethane?
7 covalent bonds
Likewise, does c2h6 have double bonds? For C2H6, carbon forms double bond with the other carbon, and each carbon has three hydrogen atoms attached to it. H-C_=C-H. Ps.
One may also ask, what type of bond does c2h6 have?
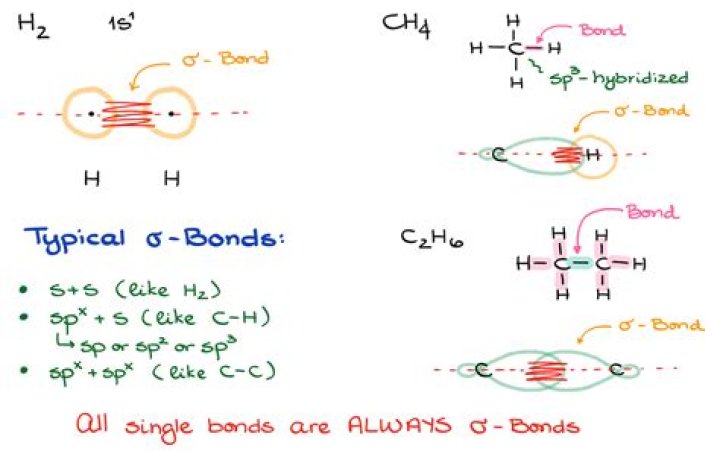
Ethane, C2H6, is a hydrocarbon molecule, an odorless gas at room temperature. The covalent bonding picture of ethane is very similar to that of methane above. Two carbon atoms, each carrying four valence electrons, come together to share a pair of electrons. We'll start calling that a single bond (or sigma bond).
Is c2h6 ionic or covalent?
You will see that, the compounds are ionic. Hydrocarbons, compounds containing only hydrogen and carbon of varying molecular size are all covalent. Examples are C2H6, C2H4, C2H2, Note that there could be some little exceptions to the examples given.
Related Question AnswersWhat kind of bond is ethane?
Ethane: The bonding in ethane is a covalent bond between carbon and hydrogen. This involves the sharing of two electrons. The two carbons in the molecule also form a single bond with each other, also a covalent bond. There is a diagram of the molecule below: Each of the bonds displayed here is a sigma bond.What is meant by covalent bond?
A covalent bond, also called a molecular bond, is a chemical bond that involves the sharing of electron pairs between atoms. These electron pairs are known as shared pairs or bonding pairs, and the stable balance of attractive and repulsive forces between atoms, when they share electrons, is known as covalent bonding.How do you find a covalent bond?
There is a couple different ways to determine if a bond is ionic or covalent. By definition, an ionic bond is between a metal and a nonmetal, and a covalent bond is between 2 nonmetals. So you usually just look at the periodic table and determine whether your compound is made of a metal/nonmetal or is just 2 nonmetals.Why Ethane is a covalent compound?
the bonds between the 2 carbon atoms and between the carbon and hydrogen atoms are single covalent bonds. the electrons are not exchanged as in the case of ionic bonds. since the electrons are shared between the atoms to satisfy the mono valency of hydrogen and tetra valency of carbon, ethane is a covalent compound.How many covalent bonds does nitrogen have?
three covalent bondsHow many covalent bonds are present in Bromoethane?
The Bromoethane molecule contains a total of 7 bond(s) There are 2 non-H bond(s). The 2D chemical structure image of Bromoethane is also called skeletal formula, which is the standard notation for organic molecules.Is ethane polar or nonpolar?
Ethane is an example of a nonpolar material. The type of bonds it has, when coupled with its shape, leaves the molecule with no regions of charge. Generally, the more carbon and hydrogen atoms a molecule has, the less polar (more nonpolar) it will be.How many covalent bonds does c2h2 molecule have?
three covalent bondsWhat type of bond is c2h4?
(A) The structural model for C2H4 consists of a double covalent bond between the two carbon atoms and single bonds to the hydrogen atoms.How do you tell if a compound is ionic or molecular?
- Mixed Ionic/Molecular Compound Naming.
- When naming compounds, the first thing you need to do is decide if the compound is ionic or molecular.
- Look at the elements in the compound.
- *Ionic compounds will contain both metals and non-metals, or at least one polyatomic ion.
- *Molecular compounds will contain only non-metals.