How many electrons are in the second energy level of an atom of each element?
.
Likewise, how many electrons are in the second energy level of an atom of each element phosphorus?
So for the element of PHOSPHORUS, you already know that the atomic number tells you the number of electrons. That means there are 15 electrons in a Phosphorus atom. Looking at the picture, you can see there are two electrons in shell one, eight in shell two, and five in shell three.
Similarly, what is the number of electrons for each element? The number of protons is the atomic number, and the number of protons plus neutrons is the atomic mass. For hydrogen, the atomic mass is 1 because there is one proton and no neutrons. For helium, it is 4: two protons and two neutrons.
2.1 Electrons, Protons, Neutrons, and Atoms.
| Element | Helium | |
|---|---|---|
| Symbol | He | |
| Atomic No. | 2 | |
| Number of Electrons in Each Shell | First | 2 |
Hereof, how many electrons are in the 2nd energy level?
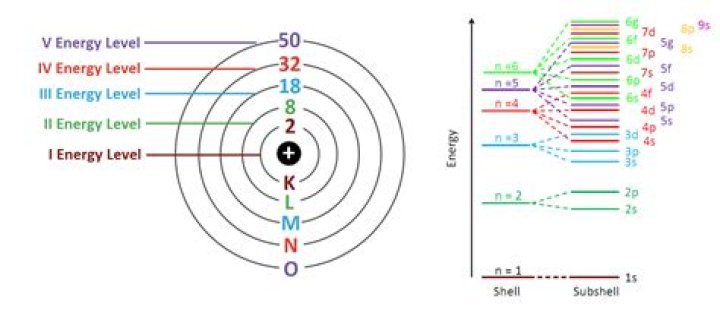
Each shell can contain only a fixed number of electrons: The first shell can hold up to two electrons, the second shell can hold up to eight (2 + 6) electrons, the third shell can hold up to 18 (2 + 6 + 10) and so on.
How many electrons are in the third energy level?
18 electrons
Related Question AnswersWhat element has 2 electrons neutral atoms?
Electrons| Atomic Number | Element | Energy Levels or "shells" |
|---|---|---|
| 1 | Hydrogen (H) | |
| 2 | Helium (He) | |
| 3 | Lithium (Li) | 1 |
| 4 | Beryllium (Be) | 2 |
How many atoms are in 1g of phosphorus?
So,6.03 x 10^23 is the number of phosphorus atoms in ONE mole.What is the number of valence electrons in phosphorus?
fiveHow many electrons are in the 2nd principle energy level N 2 of a phosphorus atom?
8 electronsWhich element contains a full second energy level?
A certain number of electrons go into a level before the next level can have electrons in it. After the first energy level contains 2 electrons (helium), the next electrons go into the second energy level. After the second energy level has 8 electrons (neon), the next electrons go into the third energy level.How do you find number of electrons in an element?
The number of electrons in a neutral atom is equal to the number of protons. The mass number of the atom (M) is equal to the sum of the number of protons and neutrons in the nucleus. The number of neutrons is equal to the difference between the mass number of the atom (M) and the atomic number (Z).What element has 16 neutrons?
phosphorusHow many atoms are in phosphorus?
Atomic mass of phosphorus is 31. But there are 4 atoms of phosphorus in one molecule of phosphorus. So mass of 1 molecule of phosphorus =(4x31)amu. = 124 amu.How many sublevels exist in the 2nd energy level?
We have s, p, d, and f sublevels. Level one has one sublevel – an s. Level 2 has 2 sublevels - s and p. Level 3 has 3 sublevels - s, p, and d.How many electrons can the 7th Shell hold?
Notice that you can easily find the number of electrons that an atom can accomodate on a particular shell, or energy level, by using the fact that each orbital can hold a maximum of two electrons.What are sublevels?
A sublevel is an energy level defined by quantum theory. In chemistry, sublevels refer to energies associated with electrons. In physics, sublevels may also refer to energies associated with the nucleus. Shell 4 can hold up to 32 electrons, Shell 5 can hold up to 50 electrons,What element contains 3 electrons in the 2p level?
| Element | Atomic number | Electron configuration |
|---|---|---|
| carbon | 6 | 1s22s22p2 |
| nitrogen | 7 | 1s22s22p3 |
| oxygen | 8 | 1s22s22p4 |
| fluorine | 9 | 1s22s22p5 |