Chemical formula: C10H8O.
Similarly one may ask, is 2 naphthol a phenol?
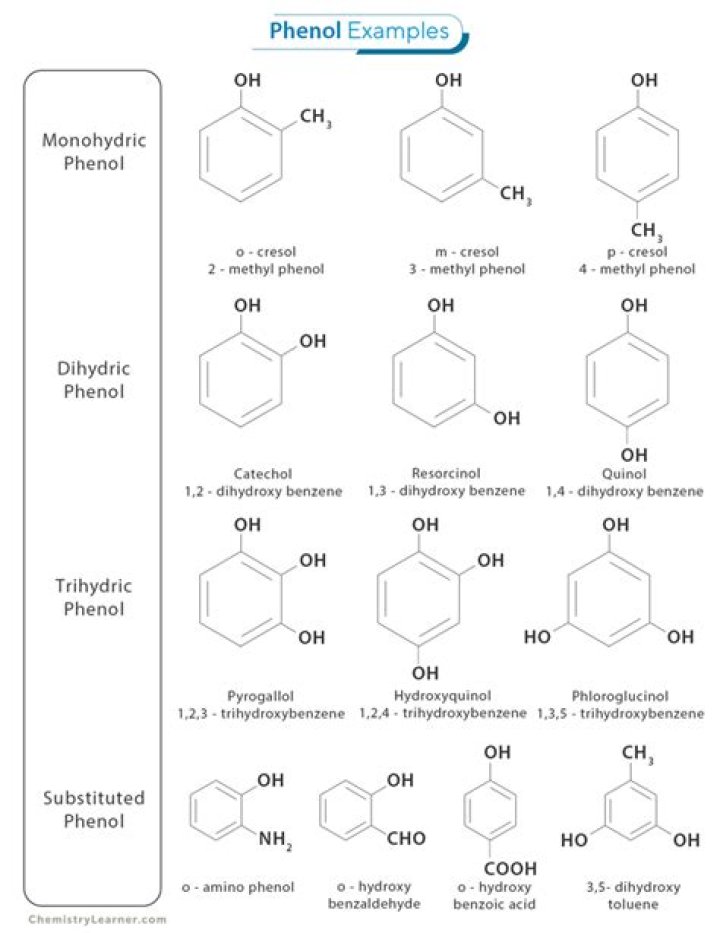
2-Naphthol, or β-naphthol, is a fluorescent colorless (or occasionally yellow) crystalline solid with the formula C10H7OH. The naphthols are naphthalene homologues of phenol, but more reactive. Both isomers are soluble in simple alcohols, ethers, and chloroform.
Additionally, what is the structure of alpha naphthol? C10H8O
Also to know, is 1 naphthol soluble in water?
1 Naphthols. The naphthols are insoluble in water. They are phenols, soluble in alkaline solution and substantive to cotton, particularly in the presence of salt. In the presence of strong alkali they are converted to the corresponding naphtholate ions and are water soluble (Fig.
Is naphthol an acid or a base?
NaOH is a strong inorganic base and both benzoic acid (a carboxylic acid) and 2-naphthol (a weak acid) are ionized to a salt in the presence of NaOH.
Related Question Answers
What does 2 naphthol smell like?
Definition for 2-naphthol (2 of 2) either of two isomeric hydroxyl derivatives, C10H7OH, of naphthalene (alpha-naphthol or 1-naphthol and beta-naphthol or 2-naphthol), white or yellowish crystals, with a phenolic odor, that darken on exposure to light: used chiefly in dyes, drugs, perfumes, and insecticides.Is phenol polar or nonpolar?
Phenol is also soluble in water to some extent. It is due to its ability to form hydrogen bonding with water molecules. However the large part of phenol molecule is phenyl group that is non polar and hence its solubility if limited in water. However the polarity of this part too increases in phenoxide ion.What is the pKa of 2 naphthol?
Figure 2. Adsorption of 2-naphthol on GCB and WSAC-3. GCB adsorbed 2-naphthol similarly from acidic and neutral solutions. This might be caused by the absence of any dissociable functional groups on both the adsorbate and adsorbent[4] in the pH range 2-7; the dissociation constant, pKa, for 2-naphthol is 9.5.What is naphthol dye?
Naphthol dyes are insoluble azo dyestuffs that are produced on the fiber by applying a Naphthol to the fiber and then combining it with a diazotized base or salt at a low temperature to produce an insoluble dye molecule within the fiber.What is the formula of beta naphthol?
C10H8O
Why does naphthol react with sodium hydroxide?
this is because Naphthol is a weak acid. for a substance to react with NaOH, it has to be acidic in nature. meaning that should be a proton donor . ethanol is an alcohol and is not acidic in nature while naphthol is acidic in nature, hence it will donate a proton to the base hence the reaction will take place.Is glucose soluble in NaOH?
Is glucose soluble in NaOH? Yes, but glucose is unstable in the presence of base and undergoes the Lobry de Bruyn–Alberda Ekenstein transformation whereby it is partially converted to a mixture of fructose and mannose.What is the melting point of beta naphthol?
2-Naphthol Properties Melting point: 120-122 °C(lit.) Boiling point: 285-286 °C(lit.)Is mothball polar or nonpolar?
Naphthalene is a non-polar molecule. This is because a bond between carbon and hydrogen has an electronegativity value of 0.4 and naphthalene consistsWhy naphthalene is aromatic?
Naphthalene has five C=C (each having two pi electrons) in the closed loop, so this molecule is aromatic. Contributor II is easily seen to have a closed loop of p orbitals hosted on a set of coplanar carbon atoms, and to obey Hückel's rule (n=2). Contributor II is aromatic, so naphthalene is aromatic.Is ethanol soluble in water?
Ethanol has a polar –OH group, which hydrogen bonds to water; which makes ethanol soluble. Ethane which consists of Ions of carbon and hydrogen only has no polar group and is not water-soluble.Is beta naphthol soluble in NaOH?
2-naphthol is slightly soluble in water, but dissolves more readily in a basic solution because it is a weak acid. The sodium hydroxide also helps to neutralize the acidic byproduct of the reaction (as H+ is displaced from the ring).Can 2 naphthol hydrogen bond with water?
Although the host framework formed by cefadroxil is very different, the remaining cavities are quite similar, because the same guest molecule can be accommodated. In all types of structures, β-naphthol is hydrogen bonded to a water molecule.Is benzoic acid soluble in NaOH?
Benzoic is soluble in a solution of NaOH because the base forms the sodium salt with the acid to form sodium benzoate. The sodium salt is soluble.Is 2 naphthol soluble in HCl?
Because this was a solubility test, 2-naphthol(brown looking) didn't dissolve in HCl.What is the composition of Molisch reagent?
Molisch reagent is made up of alpha-naphthol (C10H8OH), which is dissolved in ethanol (C2H5OH). It is used to test the presence of carbohydrates.Why is phenol more soluble than naphthol?
why phenol is so much more soluble than naphthol. The result is that the naphthol molecule does not dissolve as well in water as does phenol.Why Alpha naphthol is used in Molisch test?
Molisch test is a chemical test to detect carbohydrates. Carbohydrates undergo dehydration when heated with concentrated H2SO4 to form furfural derivatives. Furfural derivatives so obtained are condensed with alpha naphthol to give coloured compounds and hence presence of carbohydrate is confirmed.What is the difference between alpha naphthol and beta naphthol?
Alpha naphthol which is also known as the 1-naphthol is a fluroscent organic chemical substance. It's chemical formula is C10H7OH. It is the isomer of 2-naphthol. Beta naphthol which is also known as the 2-naphthanol is a crystalline organic chemical substance.