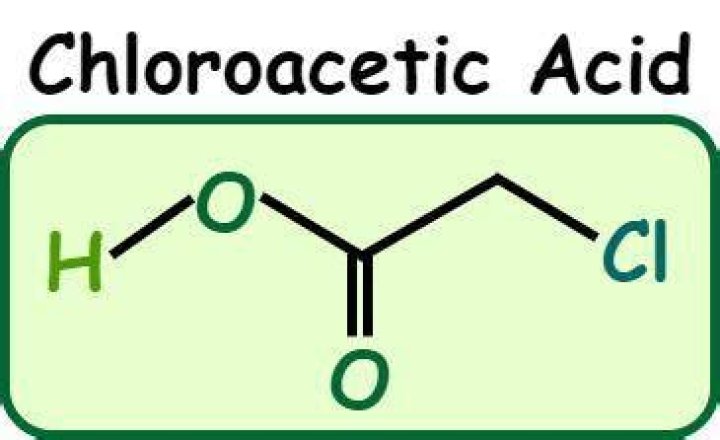
chloroacetic acid, ClCH2 COOH, in which the strongly electron-withdrawing chlorine replaces a hydrogen atom, is about 100 times stronger as an acid than acetic acid, and nitroacetic acid, NO2CH2 COOH, is even stronger..
In respect to this, why is chloroacetic acid is stronger than acetic acid?
Answer and Explanation: Chloroacetic acid is stronger than acetic acid because of the electron-withdrawing effect of chlorine. This effect is caused by the electronegativity
Also Know, why benzoic acid is stronger than acetic acid? Benzoic acid is stronger than acetic acid because the electron-donating inductive effect (+I) by the alkyl group on acetic acid destabilise the conjugate base of acetic acid (i.e. the acetate ion). In contrast, benzene rings are known to be electron dense and pull electrons away from groups attached to it.
Beside above, which is more acidic acetic acid or chloro acetic acid?
Hence chloro acetic acid is more acidic than acetic acid . Chloroacetic acid has a chlorine atom attached to its beta carbon. The inductive effect of this chlorine atom reduces the electron density over the already weakened O-H bond in the carboxylic moiety (due to the presence of alpha carbonyl group).
Why does adding chlorine increase the acidity of acetic acid?
Acidity of carboxylic acids increases due to presence of electron withdrawing group/atom since the carboxylate ion (RCOO-) gets stabilised by negative inductive effect of Cl which will withdraw the negative charge on oxygen and thereby stabilize the carboxylate ion in chloro acetic acid making it more acidic than
Related Question Answers
What affects PKA?
Several structural elements of a molecule can affect pKa including: As the electronegativity of an atom increases from left to right across a row, the acidity increases. Inductive Effect - An electronegative atom will withdraw electron density, stabilizing the conjugate base. This increases the acidity of a molecule.Which one is more acidic between acetic acid and mono chloro acetic acid and why?
Acidity of carboxylic acids increases due to presence of electron withdrawing group/atom since the carboxylate ion (RCOO-) gets stabilised by negative inductive effect of Cl which will withdraw the negative charge on oxygen and thereby stabilize the carboxylate ion in chloro acetic acid making it more acidic thanWhy PKA of ClCh2COOH is lower than PKA of ch3cooh?
In the case of Chloroacetic acid the chlorine is electronegative in nature for which it doesn't donates its electron easily and hence is more acidic in nature and it is to be noted that more will be the acidic character less will be the Pka valueso Pka value of chloroacetic acid is lower than pka value of aceticHow is trichloroacetic acid obtained from acetic acid?
Acetic acid can be converted to trichloroacetic acid by it's chlorination is presence of acetic anhydride . Trichloroacetic acid , solid is a colorless crystalline solid . Formula oftrichloroacetic is C2HC1302.Which is more acidic ch3cooh or ClCh2COOH?
ClCh2COOH is more acidic than CH3COOH because of the following reasons : “Higher electronegativity of chlorine.”Why is ccl3cooh more acidic than ch3cooh?
Since the Cl atoms in CCl4COOH are more electronegative than H atoms (in CH3COOH), they pull electron density away from the central carbon atom. This causes the carbon atom to pull electron density from the oxygen in the O-H bond, which weakens the O-H bond.Which is more acidic fluoroacetic acid or chloroacetic acid?
The pKa of fluoroacetic acid is lower than that of chloroacetic acid, which is lower than that of bromoacetic acid. Fluoroacetic acid is more acidic than chloroacetic acid, which is more acidic than bromoacetic acid.Why are phenols more acidic than alcohols?
Phenol is more acidic than alcohols due to stabilisation of phenoxide ion through resonance. Presence of electron withdrawing group increases the acidity of phenol by , stabilising phenoxide ion while presence of electron releasing group decreases the acidity of phenol by destabilising phenoxide ion.Why carboxylic acids exist as dimer?
Carboxylic acids exist as dimer because the carboxylic acid has oxygen atom and hydrogen atom. The oxygen ion has a lone pair of electron. Explanation: On the other hydrogen has a free accepting orbits for electrons.Why is Methanoic acid more acidic than Ethanoic acid?
Ethanoic acid is therefore weaker than methanoic acid, because it will re-form more easily from its ions. The other alkyl groups have "electron-pushing" effects very similar to the methyl group, and so the strengths of propanoic acid and butanoic acid are very similar to ethanoic acid.Why Acetic acid is weaker than formic acid?
Out of acetic acid and formic acid, formic acid is considered stronger because the CH3 in acetic acid is electron donating. The CH3 actually contributes electron density towards the O-H bond, making it harder to remove the H, and making acetic acid a weaker acid than formic acid.What is basicity of acetic acid?
Acetic acid contains 1 replaceable hydrogen ion per molecule of the acid or you can say it produces only one hydrogen ion per molecule of the acid. Hence, the basicity of acetic acid is 1 or it is a monobasic acid.What is acetic acid used for?
Acetic acid is one of the simplest carboxylic acids. It is an important chemical reagent and industrial chemical that is used in the production of plastic soft drink bottles, photographic film; and polyvinyl acetate for wood glue, as well as many synthetic fibres and fabrics.Which is more acidic between benzoic acid and acetic acid?
Benzoic acid is the stronger acid compared to ethanoic acid. This is due to the delocalisation of the lone pair of electrons on the oxygen atom into the benzene, increasing the polarity of the -OH bond and thus makes the cleaving of the H+ ion easier. If electron density is more than the basic character is more.Is nitric acid a strong acid?
Strong acids completely dissociate into their ions in water, while weak acids only partially dissociate. The strong acids are hydrochloric acid, nitric acid, sulfuric acid, hydrobromic acid, hydroiodic acid, perchloric acid, and chloric acid.Is acetic acid stronger than benzoic acid?
Benzene is an electron withdrawing group and hence it creates an addition partial positive charge on the Oxygen Atom present in the -OH group through propagation thus increasing its electron withdrawing power. But no such effect exists in acetic acid. Hence Benzoic acid is more acidic than Acetic acid.Is phenol a strong acid?
Phenol is a very weak acid and the position of equilibrium lies well to the left. Phenol can lose a hydrogen ion because the phenoxide ion formed is stabilised to some extent. The negative charge on the oxygen atom is delocalised around the ring. The more stable the ion is, the more likely it is to form.Why is benzoic acid weak?
Being an organic acid,Benzoic acid is a weak acid. The faster the rate of leaving proton, the more strong the acid is. Due to the resonance structure of benzoic acid, power of leaving proton is restricted and that's why its ability to leave proton is restrained. So it becomes a weak acid.Which is more acidic aliphatic or aromatic acids?
when benzoic acid ionizes it forms proton and benzoate ion ( conjugate base). Dear , its true that benzoic acid is more acidic than aliphatic carboxylic acids. but benzoic acid is less acidic than Formic acid.