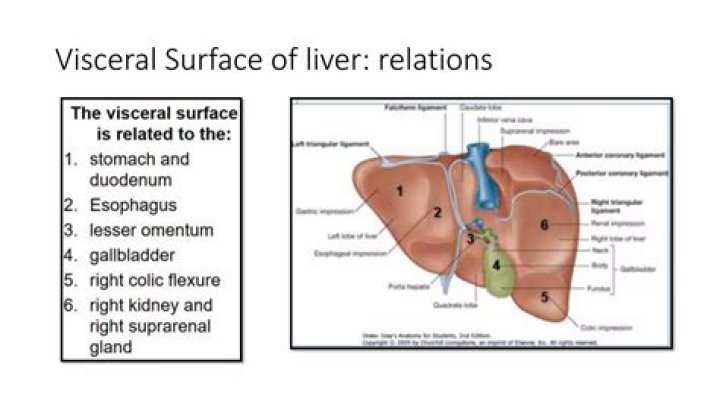
What are alkaline earth metals used in

The alkaline earth elements are found in many common materials. For example, calcium ions are a major component of your bones, magnesium is used in a variety of metal alloys, strontium is used in television tubes, and barium compounds are used as X-ray contrast agents. Radium is a very rare radioactive material.
Where are alkaline earth metals found in?
Since they have such high levels of reactivity, the alkaline earth metals never appear in their elemental state. They are often found in a compound form such as sulfates and carbonates.
Why are the two words alkaline and earth used?
Why are the two words, “alkaline” and “earth” used to name the Group IIA metals? Alkaline earth metals turn the litmus paper blue, which depicts that are basic. Alkaline earth metals are usually derived from minerals found naturally in the earth. For example, limestone.
What do alkaline earth metals tend to do?
The alkaline-earth metals tend to lose two electrons to form M 2+ ions (Be2+, Mg2+, Ca2+, and so on). These metals are less reactive than the neighboring alkali metal. Magnesium is less active than sodium; calcium is less active than potassium; and so on. These metals become more active as we go down the column.Why are they called alkali earth metals?
The name comes from the fact that the oxides of these metals produced basic solutions when dissolved in water, and they remained solids at the temperatures available to the ancient alchemists. Like the Group 1A elements, the alkaline earth metals are too reactive to be found in nature in their elemental form.
Is gold an alkaline earth metal?
BariumPlatinumGoldMercury (element)Thallium
What are the biological role of alkali and alkaline earth metals?
Calcium is used to make calcium carbonate which is used to make different products like chalk, marble, limestone etc. Alkaline earth metals release hydrogen gas when react with acids, this hydrogen gas can be used for other purposes. Alkaline earth metals are used in making electro-chemical and photoelectric cells.
What is a characteristic of alkaline earth metals?
Alkaline earth metals: A group of chemical elements in the periodic table with similar properties: shiny, silvery-white, somewhat reactive at standard temperature and pressure. They readily lose their two outermost electrons to form cations with charge +2.What are three facts about alkaline earth metals?
Interesting Facts about Alkaline Earth Metals They are called alkaline because they form solutions with a pH greater than 7, making them bases or “alkaline.” Radium is formed from the decay of uranium. It is very radioactive and is dangerous to handle. Calcium and magnesium are important for animal and plant life.
Which of the following are alkaline earth metals?Calcium (Ca), magnesium (Mg) and strontium (Sr) belong to the second group. So, they are known as alkaline earth metals.
Article first time published onWhy are alkaline earth metals good reducing agents?
Alkali metals are known as good reducing agents because they have their only one valence electron in their outermost shell. … Thus, they lose electrons and get oxidized themselves, hence reducing other compounds. Lesser the number of electrons in the valence shell, stronger will be the reducing agent.
Is Lithium alkaline earth metal?
This group includes the elements lithium, sodium, potassium, rubidium, caesium and francium. … The elements in this group, which are all shiny and silvery-white in appearance, are known as the alkaline earth metals. Like the alkali metals, they form alkaline solutions when they react with water.
What happens when alkaline earth metals react with water?
Except for beryllium, all alkaline earth metals often react with water to form highly alkaline hydroxides, which should be treated with great caution. The heavier metals of the alkaline earth react more strongly than the lighter ones. Metals react with water to create hydrogen gas and metal oxide.
What happens when alkaline earth metals are combined with hard water?
Alkaline Earth Metal Hydrides and Water Temporary hard water contains bicarbonate (HCO3-) which forms CO3-2(aq), CO2(g), and H2O when heated. The bicarbonate ions react with alkaline earth cations and precipitate out of solution, causing boiler scale and problems in water heaters and plumbing.
Can alkaline earth metals exist alone in nature?
Reactivity of Alkaline Earth Metals All alkaline Earth metals have similar properties because they all have two valence electrons. … Because of their reactivity, alkaline Earth metals never exist as pure substances in nature. Instead, they are always found combined with other elements.
Is Copper an alkali metal?
Alkali Metals are very reactive. They have low melting points and are soft enough to be cut with a knife. Potassium and sodium are two alkali metals. … This group includes iron, gold, silver, chromium, nickel, and copper, some of which are also noble metals.
What is the most common alkaline earth metals?
ABUNDANCE. Of the alkaline earth metals, calcium is the most abundant. It ranks fifth among elements in Earth’s crust, accounting for 3.39% of the elemental mass.
Is helium an alkali metal?
HydrogenSodiumPhosphorusSulfurChlorineHeliumArgon
What are the two most important alkali metals?
The most common alkali metal is sodium, which is 2.8 percent of Earth’s crust. The most common sodium compound is sodium chloride (NaCl), salt. The next most common is potassium, which is 2.6 percent of Earth’s crust. The other alkali metals are much rarer.
How many alkaline earth metals are known?
alkaline-earth metal, any of the six chemical elements that comprise Group 2 (IIa) of the periodic table. The elements are beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra).
Why are alkaline earth metals not found in nature?
-The alkali earth metals are also called s- block elements because these elements have one electron each in the valence s- subshell of their atoms. … -They are never found in nature uncombined because they are unstable and they react fast to other elements easily by losing the electron.
Can all alkali metals be cut with a knife?
Generally, all alkali metals can be easily cut with a knife. Copper, Aluminium, and Iron are hard metals with iron being the hardest amongst the given options. They cannot be cut easily with a knife as they have stronger metallic bonds.
Which metal is the strongest reducing agent?
The best reducing metal is lithium, with the maximum negative value of electrode potential. By convention, reduction potential, or the propensity to be diminished, are the normal electrode potentials.
What is the difference between an alkali metal and an alkali earth metal?
The key difference between alkali metals and alkaline earth metals is that all alkali metals have an electron in their outermost shell whereas all the alkaline earth metals have two outer electrons. … The elements in these two groups are the most reactive metals in the periodic table.
Is aluminum an alkali earth metal?
Aluminum is not an alkaline earth metal; listed in Group 13 in the periodic table, Aluminum is considered an ”Other Metal.
Why do alkali metals explode in water?
Alkali metals react with water to produce heat, hydrogen gas, and the corresponding metal hydroxide. The heat produced by this reaction may ignite the hydrogen or the metal itself, resulting in a fire or an explosion.
What elements do alkaline earth metals react with?
Except for Be, all the alkaline earth metals react with N2 to form nitrides, and all react with carbon and hydrogen to form carbides and hydrides. Alkaline earth metals dissolve in liquid ammonia to give solutions that contain two solvated electrons per metal atom.
What is the main product of reaction of an alkaline earth metal such as Ca and water?
After a second or so, the calcium metal begins to bubble vigorously as it reacts with the water, producing hydrogen gas, and a cloudy white precipitate of calcium hydroxide.
How do alkali and alkaline earth metals react with water Class 11?
The alkali metals burn vigorously in oxygen and form their respective oxides. (ii) Reactivity towards water: The alkali metals react with water and forms hydroxide and dihydrogen. Lithium due to its small size and very high hydration energy does not react with water vigorously even though it has most negative E0 value.