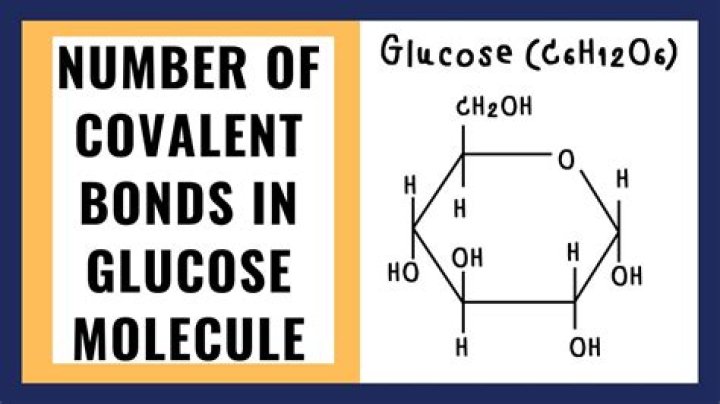
All are in a single chain. So all the bonds present in glucose is double bond and covalent bond. Double bond is present in the O atom of ketone and covalent bonds connects carbon to carbon, carbon to hydrogen and oxygen to hydrogen..
Moreover, how many bonds are there in glucose?
The carbon atoms in the glucose ring each have four covalent bonds. The best, or optimum angle, between all these bonds is 109.5o, which results in a perfect tetrahedron.
Secondly, what types of bonds are present in this molecule? Water (H2O) can be called a molecule or a compound because it is made of hydrogen (H) and oxygen (O) atoms. There are two main types of chemical bonds that hold atoms together: covalent and ionic/electrovalent bonds. Atoms that share electrons in a chemical bond have covalent bonds.
Correspondingly, does glucose contain ionic bonds?
Glucose is a covalent compound and sodium chloride is an ionic compound.
Does glucose have hydrogen bonds?
Glucose has Carbon, hydrogen and oxygen atoms. Among them, oxygen is highly electronegative and hydrogen has good electron sharing abilities. Thus, hydrogen atoms of one glucose molecules can form hydrogen bond with oxygen in water. This is what makes glucose to dissolve in water though it is a covalent molecule.
Related Question Answers
What type of bond is water?
Water is a polar molecule A water molecule is formed when two atoms of hydrogen bond covalently with an atom of oxygen. In a covalent bond electrons are shared between atoms. In water the sharing is not equal. The oxygen atom attracts the electrons more strongly than the hydrogen.What type of bond is sucrose?
Sucrose is a covalent compound. Sucrose is composed of carbon, hydrogen and oxygen, all of which have similar enough attractions for electrons to form covalent bonds with each other. A covalent bond is a bond where electrons are shared between the atoms of a compound.What type of bond is NaCl?
Ionic compounds
Is water polar or nonpolar?
Water (H2O) is polar because of the bent shape of the molecule. The shape means most of the negative charge from the oxygen on side of the molecule and the positive charge of the hydrogen atoms is on the other side of the molecule. This is an example of polar covalent chemical bonding.Is glucose a polar molecule?
The structure of glucose is as such: as can be seen, there are multiple instances of -OH groups which are highly electronegative and draw electrons away from the carbon molecules. This leads to a skewed charge distribution. It is thus polar.What type of bond is c6h12o22?
C6H12O22 is nonmetal, element 2 is nonmetal and it is a covalent bond. NaCl element 1=metal element2=non metal, it is a covalent bond type. CuZn alloy element 1=Cu=metal, element 2 is Z=m and it is metallic bond.What is the structure for glucose?
C6H12O6
Does glucose have covalent bonds?
Glucose is a covalent compound because glucose contains 3 non-metals: oxygen, carbon, and hydrogen.Is water ionic bond?
Likewise, a water molecule is ionic in nature, but the bond is called covalent, with two hydrogen atoms both situating themselves with their positive charge on one side of the oxygen atom, which has a negative charge.What type of bond is Salt?
Salt is made up of sodium and chloride and is ionically bonded. Sugar, on the other hand, is composed of carbon, oxygen, and hydrogen and has covalent bonds. A salt molecule is made up of one sodium atom and one chlorine atom. For salt to be made, the sodium atom must lose an electron and become a sodium ion.Is glucose an ion?
Since glucose is not an ionic compound, it does not dissociate, therefore i=1 (meaning one glucose molecule stays one glucose molecule).Why are ionic bonds important to life?
Ionic bonds are important because they allow the synthesis of specific organic compounds. Scientists can manipulate ionic properties and these interactions in order to form desired products. Covalent bonds are especially important since most carbon molecules interact primarily through covalent bonding.Is Wax an ionic compound?
Candle wax will melt to a liquid and burn as a gas. Only covalent compounds can be liquid or gas! Now it's time to review Ionic and Covalent compounds together! Ionic compounds are formed from a metal and a nonmetal.Is sugar a ionic or molecular compound?
Sucrose (table sugar), C12H22O11 is MOLECULAR or COVALENT compound, while sodium chloride (table salt) is _ an IONIC compound.Is NaCl ionic?
Sodium chloride (NaCl) is a typical ionic compound. The picture below shows both a sodium and a chlorine ion. Sodium has 1 electron in its outermost shell, and chlorine has 7 electrons. It is easiest for sodium to lose its electron and form a +1 ion, and for chlorine to gain an electron, forming a -1 ion.Is water a covalent bond?
H2O or water as it is more commonly known as is a molecule consisting of 2 Hydrogen molecules bonded to one Oxygen molecule. As the table shows this makes H2O a molecule with a polar covalent bond. Well, electronegativity is the measure of how attracted bond seeking electrons are to an element.Is silica ionic or covalent?
With its four valence electrons, silicon can form covalent or ionic bonds either donating or sharing its four shell electrons. At the same time, it is a relatively inert element and does not react with oxygen or water in its solid form.Why is water a polar molecule?
A water molecule, because of its shape, is a polar molecule. That is, it has one side that is positively charged and one side that is negatively charged. The molecule is made up of two hydrogen atoms and one oxygen atom. The bonds between the atoms are called covalent bonds, because the atoms share electrons.Is NaCl a molecule?
Molecules have molecular bonds. Something like table salt (NaCl) is a compound because it is made from more than one kind of element (sodium and chlorine), but it is not a molecule because the bond that holds NaCl together is an ionic bond. If you like, you can say that sodium chloride is an ionic compound.