1 Answer. Actually, water has all three types of intermolecular forces, with the strongest being hydrogen bonding. All things have London dispersion forcesthe weakest interactions being temporary dipoles that form by shifting of electrons within a molecule..
Moreover, what kinds of intermolecular forces exist for h2o?
Ernest Z. Water has hydrogen bonds, dipole-induced dipole forces, and London dispersion forces.
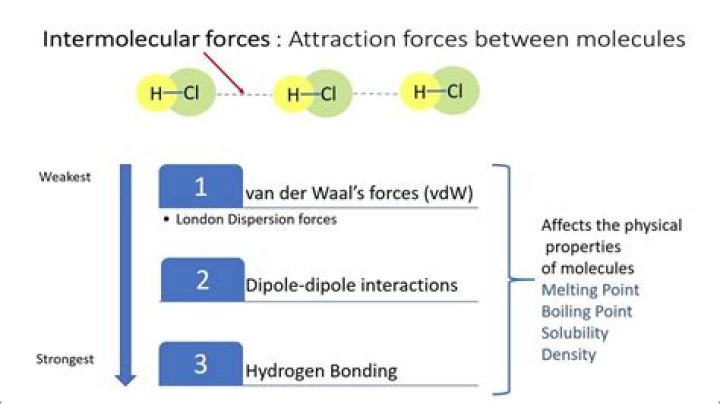
In order of decreasing strength, the types of intermolecular bonds in covalent substances are:
- Hydrogen bonds.
- Dipole-dipole attractions.
- Dipole-induced dipole attractions.
- London dispersion forces.
Subsequently, question is, what type of intermolecular forces exist between ch3oh and h2o? CH3OH, or methanol, displays London dispersion forces and hydrogen bonding. London dispersion forces are the weakest of the intermolecular forces.
Likewise, people ask, what life would be if water molecules are not held by hydrogen bonds?
For example to the boiling temperature. This extra forces holding the water molecules together means that water stays liquid at room temperature whereas other very similar molecules that don't form hydrogen bonds are gaseous. This is why life on earth is water based rather than, say, hydrogen sulfide based.
What types of intermolecular forces are present in each molecule?
Intermolecular forces act between molecules. In contrast, intramolecular forces act within molecules. Intermolecular forces are weaker than intramolecular forces. Examples of intermolecular forces include the London dispersion force, dipole-dipole interaction, ion-dipole interaction, and van der Waals forces.
Related Question Answers
Is water a dipole?
Water is a dipolar molecule because each atom has a dipole, or partial charge. Oxygen is more electronegative than hydrogen, and thus pulls the Water is a dipolar molecule because each atom has a dipole, or partial charge. These partial charges are called dipoles and cause water to be considered a dipolar molecule.Is water dipole dipole?
Water has strong hydrogen bond dipole-dipole intermolecular forces that give water a high surface tension and a high heat of vaporization and that make it a strong solvent.Is HCl dipole dipole?
HCl molecules, for example, have a dipole moment because the hydrogen atom has a slight positive charge and the chlorine atom has a slight negative charge. Because of the force of attraction between oppositely charged particles, there is a small dipole-dipole force of attraction between adjacent HCl molecules.Is h2s a dipole dipole?
H2S, H2Se and H2Te exhibit dipole-dipole intermolecular forces while H2O exhibits hydrogen bonding. C4H10 is a nonpolar hydrocarbon molecule so has the dispersion force (42 electrons) and has a stronger force of attraction than CO2 (bp −0.5°C).Is h2o hydrogen bonding?
In H2O molecule, two water molecules are bonded by a Hydrogen bond but the bond between two H - O bonds within a water molecule are covalent. The dotted lines represent a hydrogen bond and the solid lines represent a covalent bond.What is the strongest intramolecular force?
Dipole-dipole interactions are the strongest intermolecular force of attraction.What is the strongest intermolecular force?
hydrogen bonding
Is h2o polar or nonpolar?
A water molecule, abbreviated as H2O, is an example of a polar covalent bond. The electrons are unequally shared, with the oxygen atom spending more time with electrons than the hydrogen atoms. Since electrons spend more time with the oxygen atom, it carries a partial negative charge.What type of bond is water?
Water is a polar molecule A water molecule is formed when two atoms of hydrogen bond covalently with an atom of oxygen. In a covalent bond electrons are shared between atoms. In water the sharing is not equal. The oxygen atom attracts the electrons more strongly than the hydrogen.Why are hydrogen bonds in water important?
Hydrogen bonds in water provide many characteristic benefits to water: cohesion (holding water molecules together), high specific heat (absorbing heat when breaking, releasing heat when forming; minimizing temperature change), high heat of vaporization (several hydrogen bonds must be broken in order to evaporate water)Why are hydrogen bonds weak?
Hydrogen bonds are relatively weak, but they add to the energy needed for molecules to move apart from each other when matter changes state from a solid to a liquid or from a liquid to a gas. This explains why polar covalent compounds have relatively high melting and boiling points.Why are hydrogen bonds important?
Hydrogen bonding is important in many chemical processes. Hydrogen bonding is responsible for water's unique solvent capabilities. Hydrogen bonds hold complementary strands of DNA together, and they are responsible for determining the three-dimensional structure of folded proteins including enzymes and antibodies.Is water a covalent bond?
H2O or water as it is more commonly known as is a molecule consisting of 2 Hydrogen molecules bonded to one Oxygen molecule. As the table shows this makes H2O a molecule with a polar covalent bond. Well, electronegativity is the measure of how attracted bond seeking electrons are to an element.Why are water molecules attracted to each other?
The oxygen-hydrogen (O–H) bonds in water make it a polar molecule. That means the water molecule has an area of positive charge and an area of negative charge. This polarity makes water molecules attracted to each other.How do hydrogen bonds between water molecules occur?
Hydrogen-bonding forms in liquid water as the hydrogen atoms of one water molecule are attracted towards the oxygen atom of a neighboring water molecule; generally, a proton shared by two lone electron pairs. Hence, the oxygen atom is partially negatively charged, and the hydrogen atom is partially positively charged.Why is water a polar molecule?
A water molecule, because of its shape, is a polar molecule. That is, it has one side that is positively charged and one side that is negatively charged. The molecule is made up of two hydrogen atoms and one oxygen atom. The bonds between the atoms are called covalent bonds, because the atoms share electrons.Is c2h5oh dipole dipole?
C2H5F is a polar molecule and is held together by dipole interactions, which are generally stronger than dispersion forces. C2H5OH has hydrogen bonding which is stronger than either ordinary dipole interaction and dispersion force.Is methanol a dipole dipole force?
Methanol is not an ionic molecule and will not exhibit intermolecular ionic bonding. Methanol is polar, and will exhibit dipole interactions. It also contains the -OH alcohol group which will allow for hydrogen bonding.What is the strongest intermolecular force in ch3oh?
Hydrogen bonding