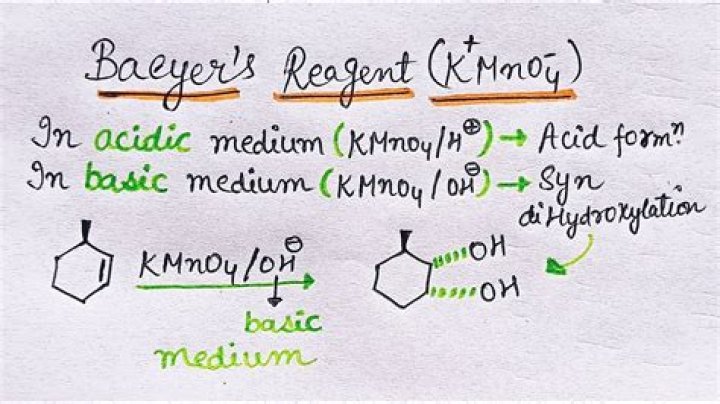
Baeyer's reagent is an alkaline solution of cold potassium permanganate, which is a powerful oxidant making this a redox reaction. Reaction with double or triple bonds (-C=C- or -C≡C-) in an organic material causes the color to fade from purplish-pink to brown. It is a syn addition reaction..
Keeping this in view, what is meant by Baeyer's reagent?
Baeyer's reagent is an alkaline solution of cold potassium permanganate, which is a powerful oxidant. It dissolves in water to give intensely pink or purple solutions. On evaporation it leaves prismatic purplish-black glistening crystals.
One may also ask, what happens when ethene is treated with Baeyer's reagent? By adding cold dilute Baeyer's reagnant If this mixture is added to an unsaturated compounds such as alkenes or alkanes then : Purple colour of the reagnant changes .
Furthermore, how do you make baeyer's reagent?
To obtain Baeyer's reagent the solution is transferred to a dark-colored glass bottle, 10 grams of anhydrous sodium carbonate () is added to the solution, and the stoppered bottle is shaken until fully dissolved. This creates the reagent, which consists of alkaline potassium permanganate.
What does a positive baeyer test look like?
The Baeyer test reagent is a very deep purple color. In a positive Baeyer test the deep purple color is replace with a brown precipitate. The common functional groups which give a positive Baeyer test are alkenes and alkynes. In a negative Baeyer test, the purple color is retained and no brown precipitate is observed.
Related Question Answers
What is baeyer's reagent formula?
Answer: Baeyer's reagent is an alkaline solution of cold potassium permanganate, which is a powerful oxidant making this a redox reaction. Reaction with double or triple bonds (-C=C- or -C≡C-) in an organic material causes the color to fade from purplish-pink to brown.What does Schiff's test for?
The Schiff test is an early organic chemistry named reaction developed by Hugo Schiff, and is a relatively general chemical test for detection of many organic aldehydes that has also found use in the staining of biological tissues.How do you test for aldehydes?
To carry out the test, you add a few drops of the aldehyde or ketone to the freshly prepared reagent, and warm gently in a hot water bath for a few minutes. No change in the colourless solution. The colourless solution produces a grey precipitate of silver, or a silver mirror on the test tube.What is being oxidized in a baeyer test?
The Bayer's test uses for dilute Potassium Permaganate to oxidised double bonded alkenes or triple bonded alkynes , called oxidation because alkene replaced by -OH groups and charge goes from +1 oxidation state to +2 oxidation state , it means there is lossing 1 electron means there is oxdaion….What is ozonolysis reaction?
Ozonolysis is an organic reaction where the unsaturated bonds of alkenes, alkynes, or azo compounds are cleaved with ozone. Alkenes and alkynes form organic compounds in which the multiple carbon–carbon bond has been replaced by a carbonyl group while azo compounds form nitrosamines.Why do alkenes Decolourise bromine water?
?Therefore, when alkenes come into contact with bromine water, they cause it to decolorize. bromine water is commonly used to test for the presence of an alkene which contains a double covalent bond which reacts with the bromine water which changes its color from an intense yellow to a colorless solution.Why the Colour of Baeyer's reagent gets discharged when treated with an alkene?
Bayer reagent is an Alkaline solution cold of potassium permanganate primarily act as a powerful oxidant. When Bayer reagent comes to contact with an alkene, the alkene is oxidized to glycol which is the reason behind the discharged color of Bayer reagent whereas permanganate reduced to Mno2 which gives Brown color.What is bromine water test?
bromine water test is a test for making a comparative study of an alkane or alkene. Alkene on one hand would react with the bromine water and would leave it decolourised whereas alkane on other hand would not react with it and leaves bromine water orange coloured(it's original colour). 186 views.Is KMnO4 an oxidizing agent?
Potassium Permanganate ? Potassium permanganate is an inorganic chemical compound with the formula KMnO4. It is a salt consisting of K+ and MnO4 − ions. Formerly known as permanganate of potash or Condy's crystals, it is a strong oxidizing agent. It dissolves in water to give intensely purple solutions.Why KMnO4 is Coloured?
Potassium permanganate(KMnO4) is colored because it absorbs light in the visible range of the electromagnetic spectrum. The permanganate ion is the source of the color, as a ligand-to-metal charge transfer takes place between the oxygen's p orbitals and the empty d-orbitals on the metal.Why KMnO4 is a self indicator?
So once all the permanganate ions are used up in the reaction, the solution loses its pink colour. This indicates the end of the reaction and hence potassium permanganate is called a self indicator as it acts as an indicator apart from being one of the reactants.Is KMnO4 polar or nonpolar?
Potassium permanganate is an Ionic molecule. Polar or non-polar usually refers to covalent molecules. The potassium has given it's electron to the permanganate polyatomic Ion, so it is more appropriate to ask about it. Potassium would be K+.What does potassium permanganate do to skin?
When applied to your skin, potassium permanganate kills germs by releasing oxygen when it meets compounds in your skin. It also acts as an astringent, which is a drying agent. Some of the conditions that potassium permanganate can help treat include: Infected eczema.Is KMnO4 carcinogenic?
CARCINOGENIC EFFECTS: Not available. MUTAGENIC EFFECTS: Mutagenic for bacteria and/or yeast. TERATOGENIC EFFECTS: Not available. DEVELOPMENTAL TOXICITY: Not available.Can we drink potassium permanganate water?
Potassium permanganate oxidizes iron, manganese, and hydrogen sulfide into particles. When treating water to remove iron bacteria, a solution of potassium permanganate is fed into the well. A concentration of 3.8 to 7.6 grams per gallon has been found to be very effective.Is KMnO4 a base?
- Salts from strong bases and weak acids: pH greater than 7 (alkaline). Based on these rules, the solution of KMnO4 dissolved in water is Basic.How do you test for unsaturation?
Bromine Test In this test when bromine solution is added to the unsaturated hydrocarbon the brown colour disappears if the hydrocarbon is unsaturated. Bromine forms an addition product with the unsaturated hydrocarbon. Note: Decolourization of orange-red colour of bromine solution indicates unsaturation.What happens when ethene reacts with KMnO4?
If KMnO4/H reacts with ethene, what equation will it give? KMnO4 racts with double bond to oxidise it to Cis-diol and then further oxidise the C-C bond to carbonyl conpound (if adehyde formed then it oxidises to corresponding carboxylic acid). This formaldehyde will further oxidise to formic acid.What happens when ethylene is treated with alkaline KMnO4?
When ethylene is passed through alkaline KMnO4 solution 1, 2-Ethanediol is formed. The Purple color of KMnO4 decolorizes.