Diethyl ether, or simply ether, is an organic compound in the ether class with the formula (C..
Similarly, what is ether made out of?
Ethyl ether, also called diethyl ether, well-known anesthetic, commonly called simply ether, an organic compound belonging to a large group of compounds called ethers; its molecular structure consists of two ethyl groups linked through an oxygen atom, as in C2H5OC2H5.
Subsequently, question is, is ch3ch2och2ch3 polar or nonpolar? Plan: CH3CH2OCH2CH3 is polar with dipole-dipole interactions as the dominant intermolecular forces. Examine the solutes to determine which has intermolecular forces more similar to those in diethyl ether. This solute is the one that would be more soluble.
Besides, is diethyl ether illegal?
Ether is used as a solvent by scientists for chemical reactions. People used to take ether as a recreational drug (drugs used for fun, such as alcohol) in the 1800s. It became illegal to sell ether in Poland in 1923. Ether is made as a byproduct when ethanol is made, so it is not very expensive.
Is ether still used today?
Usage of ether and chloroform later declined after the development of safer, more effective inhalation anesthetics, and they are no longer used in surgery today.
Related Question Answers
When did Ether stop being used?
The first true demonstration of ether as an inhalation anesthetic was on October 16, 1846 by William T.G. Morton, a Boston dentist. Ether has been replaced completely by newer inhalation agents and open drop delivery systems have been exchanged for complicated vaporizers and monitoring systems.How long does ether high last?
How long does the feeling last? Several breaths of solvents will produce a high within a few minutes of use. This high may last up to 45 minutes if no more breaths are taken.Is ether an element?
The element ether, called “akasha” in Sanskrit is the first of the five great elements (pancha mahabhutus). It comes first because it is the most subtle of the elements. Often referred to as “space,” it is the essence of emptiness. It is the space the other elements fill.Can you make ether?
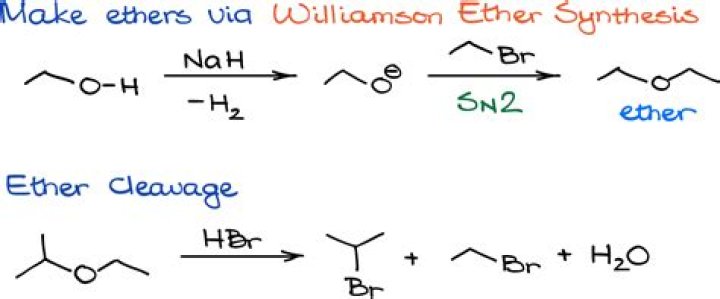
Ethers can be prepared by a method called the Williamson ether synthesis. In this reaction a halide ion is displaced from an alkyl halide by an alkoxide ion in an SN2 reaction. The alkoxide ion is prepared by the reaction of an alcohol with a strong base such as sodium hydride.Is ether an acid or base?
Ethers serve as Lewis bases and Bronsted bases. Strong acids protonate the oxygen to give "oxonium ions".Is ethanol flammable?
Ethanol, also known as ethyl alcohol, is a colorless flammable liquid that has a pungent taste. Produced by fermentation, it is the alcohol found in alcoholic beverages and used as a solvent. Ethanol, C2H5OH or C2H6O, is flammable, colorless, and slightly toxic.What are ethers used for?
Ethers are liquid at room temperature and are typically colorless, with a sweet smell. The most common type of ether is diethyl ether, which is extremely flammable and was one of the first anesthetics used in surgery. Due to its anesthetic effects, ether is also used as an illicit drug to induce sedation and euphoria.Is ether an alcohol?
Ether, any of a class of organic compounds characterized by an oxygen atom bonded to two alkyl or aryl groups. In an alcohol one hydrogen atom of a water molecule is replaced by an alkyl group, whereas in an ether both hydrogen atoms are replaced by alkyl or aryl groups.Is ether a drug?
The anesthetic and intoxicating effects of ether have made it a recreational drug. Diethyl ether in anesthetic dosage is an inhalant which has a long history of recreational use. In the 19th century and early 20th century ether drinking was popular among Polish peasants.How bad is ether for you?
Acute: harmful by inhalation in high concentrations which can cause inebriation, sedation, unconsciousness and respiratory paralysis. Diethyl ether is irritating to the eyes, respiratory system and skin but these effects are usually reversible on removal of exposure.Can you purchase ether?
Unfortunately, there is no easy way to buy Ether with PayPal. You will first have to buy bitcoins with PayPal on VirWoX. Once you've purchased bitcoins there you can use an exchange like Changelly or Binance to exchange your bitcoins for Ether.Is ether soluble in water?
Water is polar in nature while Ether is non-polar. Ethers are very slightly soluble in water at around 6.8g/100g. This is due to the presence of oxygen in ethers which interact with hydrogen in water to produce H-bonding to make it slightly soluble.Is ethanol miscible in water?
Ethanol has a 2 carbon chain and a OH group. As water is polar it attracts OH group. Because of the strength of the attraction of the OH group, first three alcohols (methanol, ethanol and propanol) are completely miscible. They dissolve in water in any amount.Why is ch3coch3 polar?
The oxygen linked to the carbon has a tendency to attract the pair of bond electrons between the oxygen and carbon, so CH3COCH3 is polar.Is ether a polar or nonpolar solvent?
Ethers are considered (somewhat) polar aprotic solvents, because — while they cannot donate hydrogen bonds — they can accept them, and also are able to solvate cations but not anions.Is co2 polar or nonpolar?
Carbon dioxide is non-polar because of the symmetry of its bonding. The electronegativity difference between carbon and oxygen is 1.0, which makes the bonds polar. However, the two polar bonds are at 180 degrees to each other so the dipoles cancel out.Why is dichloromethane polar?
The individual bonds within dichloromethane are somewhat polar due to the electronegativity differences between C&H and between C&Cl, so yes, it is polar at the bond level. The C-Cl bonds are more polar than the C-H bonds and so there is some net residual polarity.When did ether start being used?
October 16, 1846