212 °F
.
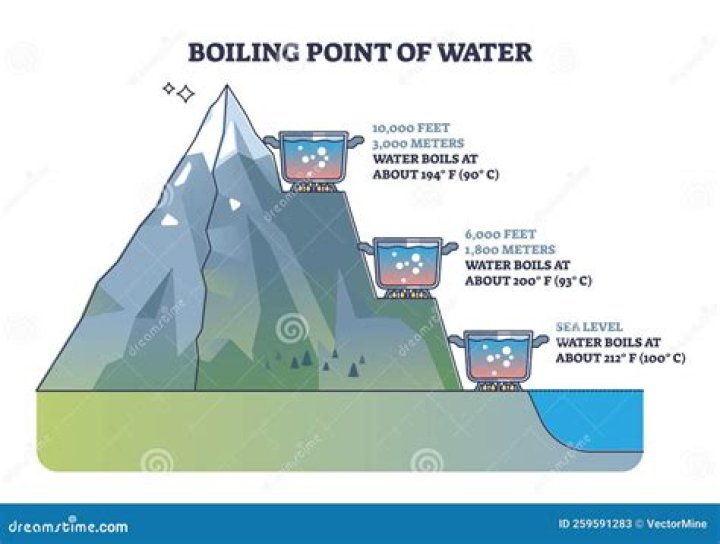
Moreover, what happens to boiling point of water at high altitudes?
At higher altitudes, air pressure is lower. When atmospheric pressure is lower, such as at a higher altitude, it takes less energy to bring water to the boiling point. Less energy means less heat, which means water will boil at a lower temperature at a higher altitude.
Similarly, what is the boiling point of water at an elevation of 6500 ft? Boiling Point of Water at Different Altitudes
| Altitude ft. (meters) | Boiling Point - Fahrenheit | Boiling Point - Celsius |
| 6000 ft. (1829 m.) | 200.5 ºF | 93.5 ºC |
| 6500 ft. (1981 m.) | 199.5 ºF | 93 ºC |
| 7000 ft. (2134 m.) | 198.5 ºF | 92.5 ºC |
| 7500 ft. (2286 m.) | 198 ºF | 92 ºC |
In this manner, does it take water longer to boil in high altitude?
At a higher elevation, the lower atmospheric pressure means heated water reaches its boiling point more quickly—i.e., at a lower temperature. This is the opposite of what many people suppose: that water takes longer to boil on high. As we've just demonstrated, boiling water at altitude is quicker.
Why does water boil at less than 100 at high altitude?
The boiling of water depends on the atmospheric pressure at that altitude. At high altitudes, the pressure is lower, so water cannot boil at 100 degrees. The boiling point of water at a high altitude is less than 100 degrees.
Related Question Answers
Can water boil 0 Celsius?
Answer: you can boil water at 0 degrees as it depends on temperature as well as pressure. In fact pressure decides the boiling temperature of water, for example at 1 atmospheric pressure,100 degrees is the temperature at which the water starts boiling. It may be around 0.00603 atmospheric pressure.What lowers the boiling point of water?
Sugar, salt or other non-volatile solutes in water will usually make the boiling point higher. Alcohol, in contrast, is a volatile chemical that lowers the boiling point of water. Even a large amount dissolved in the water will usually make only small changes in the boiling point.How long boil potatoes high altitude?
At altitudes below 1,000 feet, boil foods for 10 minutes. Add an additional minute of boiling time for each additional 1,000 feet elevation (for example, at 3,000 feet, boil for 12 minutes).Does water boil faster with salt?
Salt increases water's boiling point, or the temperature it must reach in order to boil. This new salt water solution needs more heat to start boiling than pure water does, so the time it takes to boil increases slightly. So your water is hotter, but it's not boiling any faster.What is the boiling point of pure water?
100 degrees Celsius
What do you mean by altitude?
As a general definition, altitude is a distance measurement, usually in the vertical or "up" direction, between a reference datum and a point or object. The reference datum also often varies according to the context.Does water always boil at 100 degrees Celsius?
Introduction. We all learn at school that pure water always boils at 100°C (212°F), under normal atmospheric pressure. And removing dissolved air from water can easily raise its boiling temperature by about 10 degrees centigrade.What happens to the boiling point of water at high altitudes quizlet?
How does a higher altitude affect boiling point? Atmospheric pressure is lower at high altitudes and the temperature is lowered for boiling. Because they dilute the water molecules and lower vapor pressure.How long boil eggs high altitude?
Bring a large pot of water to a boil. (Make sure you have enough water to cover the eggs completely and that the pot is big enough for the eggs to be in a single layer.) Using a large spoon, slowly and carefully lower the eggs down into the boiling water. Cook eggs for 15 minutes.Which force makes it harder to boil water?
Hydrogen bonding is between hydrogen atom of one water molecule and oxygen atom of another water molecule (H2O - - - - H2O). Though its a weak force of attraction, energy is needed to break these bonds. Collectively this energy is considerable and so the boiling point of H2O is 100 degree Celsius.Does water freeze faster at higher altitude?
In an ordinary container (exposed to atmospheric pressure), yes. The freezing point will increase with altitude (and corresponding decrease in atmospheric pressure), but the difference is very slight.Why does less water boil quicker?
Originally Answered: Why does less water boil faster than more water? Simple: you're putting a constant flow of energy into a smaller quantity of water, which means that its temperature rises more quickly than the same amount of energy (per minute) being applied to a larger quantity.Why does the boiling point decrease as altitude increases?
As elevation increases, atmospheric pressure decreases because air is less dense at higher altitudes. Because the atmospheric pressure is lower, the vapour pressure of the liquid needs to be lower to reach boiling point. The boiling point is lower at higher altitude.What is boiling point of water in pressure cooker?
121 °C
Can water temperature exceed boiling point?
Liquid water can be hotter than 100 °C (212 °F) and colder than 0 °C (32 °F). Heating water above its boiling point without boiling is called superheating. If water is superheated, it can exceed its boiling point without boiling. To experience this, put a container of bottled water into a bowl of ice.Can you give reason why it takes longer time to cook at high altitude?
At higher altitudes, say like mountains, cooking food is difficult. Higher altitudes means lower atmospheric pressure and thus lower boiling point. So, at higher altitude, food needs to be cooked for a longer time due to the lower in boiling point of water.Why does water boil in a vacuum?
Lack Of Atmospheric Pressure? In a vacuum, there is no pressure. This is critical for most liquids to remain in a liquid state because with no pressure, the temperature at which they start to boil drops. Water boils when there is no pressure (and as morbid as it sounds, so does blood).How do you calculate the boiling point of water?
Solving for Boiling Point 045°C/mmHg. Simplified, 100°C=BPobs+7.2. The units of mmHg cancel each other out, leaving the units as degrees Celsius. Solved for the boiling point at 600mmHg, the equation becomes: BPobs=100°C-7.2°C=92.8°C.Does salt decrease the boiling point of water?
Effect of Salt on Boiling Water Adding salt does not lower the boiling point of water. Actually, the opposite is true. Adding salt to water results in a phenomenon called boiling point elevation. The boiling point of water is increased slightly, but not enough that you would notice the temperature difference.