The PH3 bond angle will be about 90 degreessinceit has a trigonal pyramidal molecular geometry (it will be abitless since the lone pair will push down)..
Beside this, what is the ideal bond angle in ph3?
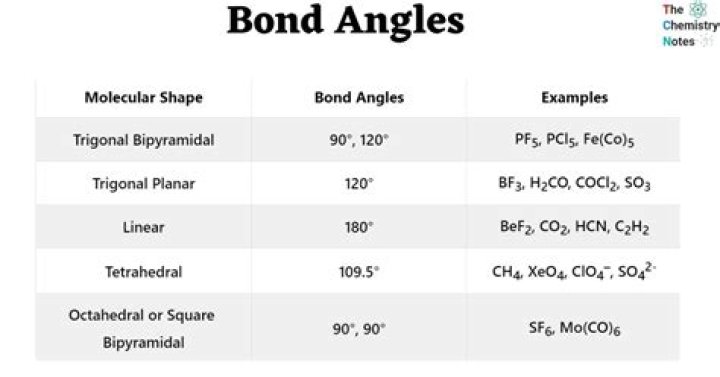
In ammonia sp3 hybridization occurs .So itsbondangle should have been 109°28' . But due to thepresence oflone pair it is slightly less. So it was expected thatthe anglein PH3 will also be approximately same. But , itwas found tobe around 90°.
Similarly, what is the shape of ph3? In case of PH3, ans is sp3 with one lone pairsoit is pyramidal shaped. .
Simply so, what is the bond angle of phosphine?
Structure of Phosphine The bond angle H-P-H is = 93°. On theotherhand, ammonia has pyramidal geometry with a bond angleof107.80. Thus, we see that both these chemicals haveacomparative bond angle. Phosphorus is lesselectronegativethan nitrogen.
Why bond angle of pf3 is greater than ph3?
While the bond angle of PH3 islessthan that of PF3. Both PH3 and PF3arepyramidal in shape. The bond angle is more in PF?3 duetolone pair-bond pair repulsion. As flourine is smallerinsize and more electronegative than hydrogen, thebondpair of electrons is towards F.
Related Question Answers
Is ph3 trigonal planar?
According to VSEPR theory, the geometry of thePH3molecule is best described as: linear, trigonalplanar,tetrahedral, bent, or trigonalpyramidal.Is nf3 tetrahedral?
In NF3 there are also three bond pairs, butthenitrogen has a lone pair as well. The four pairs ofelectronsarrange themselves tetrahedrally, but thedescription of theshape only takes account of the atoms. NF3is pyramidal. Inmethane, the four bond pairs get as far apart aspossible in atetrahedral arrangement.Is ph3 trigonal pyramidal?
The molecular geometry of PH3 istrigonalpyramidal.Is h2o polar or nonpolar?
A water molecule, abbreviated as H2O, isanexample of a polar covalent bond. The electronsareunequally shared, with the oxygen atom spending more timewithelectrons than the hydrogen atoms. Since electrons spend moretimewith the oxygen atom, it carries a partialnegativecharge.Is ph3 a polar molecule?
Phosphine is an example of a polar moleculewithnon-polar bonds. It has the formula PH3. Thelonepair of electrons on phosphorus causes the molecule tobeasymmetrical with respect to charge distribution and that's whythemolecule is polar even though it hasnon-polarbonds. Its shape is trigonalpyramidal.Is h2s polar or nonpolar?
Hydrogen sulfide is nonpolar. Even thoughthemolecular geometry would allow for it to polar, thebondsare not polar, so the molecule isn't either. Polarityisdetermined by electronegativity.How many lone pairs are in ph3?
(b) PH3: Phosphorus (Group 5A) has fivevalenceelectrons, and will complete its octet by making threebonds. Thereare three hydrogens, so it makes a single bondwitheach.What kind of bond is ph3?
PH3 is a covalent polar compound. Phosphorousisbonded to three hydrogen atoms and has a lone pair ofelectrons.Since the electronegativity of phosphorous and hydrogenis nearlysame so the covalent bond isnon-polar.What shape is becl2?
The molecular geometry of BeCl2 is linearwithsymmetric charge distribution around the centralatom.Is nh3 bent or linear?
If these are all bond pairs the molecular geometryistetrahedral (e.g. CH4). If there is one lone pair of electronsandthree bond pairs the resulting molecular geometry istrigonalpyramidal (e.g. NH3). If there are two bond pairsand twolone pairs of electrons the molecular geometry is angularorbent (e.g. H2O).Which has greater bond angle ph3 or pf3?
PH3 and PF3 are also pyramidal inshapewith one lone pair on P. But PF3 has greater bond anglethanPH3. In PF3 the lone pair on the phosphoruspushesthe P-F bonding electrons away from itself, andresulting inresonance, leading to partial doublebondcharacter.Which has greater bond angle nh3 or ph3?
In NH3, there are 4 electron pairs(3bonding pairs & 1 lone pair) in the outer most shellofN. The repulsion between lone pair and a bond pairofelectrons always exceeds to that of two bond pairs.Thus,the bond angle of PH3 molecule is lesser thanthat inNH3 molecule.