What is the difference between enthalpy and molar enthalpy?
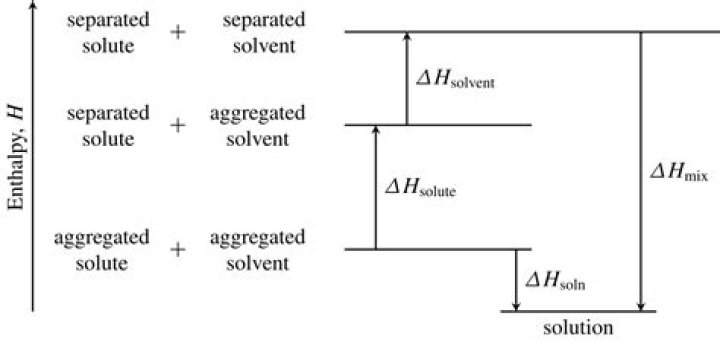
.
Simply so, what is the difference between enthalpy and standard enthalpy?
Remember that an enthalpy change is the heat evolved or absorbed when a reaction takes place at constant pressure. The standard enthalpy change of a reaction is the enthalpy change which occurs when equation quantities of materials react under standard conditions, and with everything in its standard state.
Also Know, what is enthalpy in simple terms? Enthalpy. When a substance changes at constant pressure, enthalpy tells how much heat and work was added or removed from the substance. Enthalpy is similar to energy, but not the same. When a substance grows or shrinks, energy is used up or released.
Also Know, what is molar enthalpy?
Molar enthalpy of vaporization is the amount of energy needed to change one mole of a substance from the liquid phase to the gas phase at constant temperature and pressure. The usual unit is kilojoules per mole (kJ/mol).
How do you find enthalpy?
Use the formula ∆H = m x s x ∆T to solve. Once you have m, the mass of your reactants, s, the specific heat of your product, and ∆T, the temperature change from your reaction, you are prepared to find the enthalpy of reaction. Simply plug your values into the formula ∆H = m x s x ∆T and multiply to solve.
Related Question AnswersHow do I calculate entropy?
But entropy change is quoted in energy units of J. That means that if you are calculating entropy change, you must multiply the enthalpy change value by 1000. So if, say, you have an enthalpy change of -92.2 kJ mol-1, the value you must put into the equation is -92200 J mol-1.What are the different types of enthalpy?
Types of Enthalpy Change- Heat of formation. The enthalpy change that takes place when one mole of compound is formed from its elements.
- Heat of combustion. The enthalpy change that takes place when one mole of compound is completely burnt in excess of air or oxygen.
- Heat of neutralization.
- Heat of solution.
What affects enthalpy of combustion?
Enthalpy change of combustion is the energy released when 1 mole of a compund is burned in excess of oxygen to form products at 298k and 1 atm pressure or under standard conditions. Example: C2H6(g) + 7/2O2 -> 2CO2(g) +3H20(l) NOTE: Compounds in stable state (room temperature, 298k 1 atm pressure)What is Delta H degree?
The standard enthalpy of formation (Delta H degree f) is the enthalpy change that occurs when exactly 1 mol of a compound is formed from its constituent elements under standard conditions. The standard conditions are 1 atm pressure, a temperature of 25 degree C, and all the species present at a concentration of 1 M.What does Delta H mean?
In chemistry, the letter "H" represents the enthalpy of a system. Enthalpy refers to the sum of the internal energy of a system plus the product of the system's pressure and volume. The delta symbol is used to represent change. Therefore, delta H represents the change in enthalpy of a system in a reaction.What is Hess's Law equation?
The enthalpy change for the overall process is the sum of the enthalpy change of the steps in the process. This is known as Hess's Law and is given in the following equation. ΔHrxn=ΔH1+ΔH2+ΔH3+⋯What does negative molar enthalpy mean?
It means that the system in which the chemical reaction is occurring is gaining energy. If the enthalpy change of a reaction is negative, the system is losing energy, so the products have less energy than the reactants, and the products are lower on the vertical energy scale than the reactants are.How do you solve molar enthalpy problems?
Enthalpy of Solution (Heat of Solution) Example- Calculate the heat released, q, in joules (J), by the reaction: q = mass(water) × specific heat capacity(water) × change in temperature(solution)
- Calculate the moles of solute (NaOH(s)): moles = mass ÷ molar mass.
- Calculate the enthalpy change, ΔH, in kJ mol-1 of solute:
How do you determine molar mass?
Key Points- The molar mass is the mass of a given chemical element or chemical compound (g) divided by the amount of substance (mol).
- The molar mass of a compound can be calculated by adding the standard atomic masses (in g/mol) of the constituent atoms.