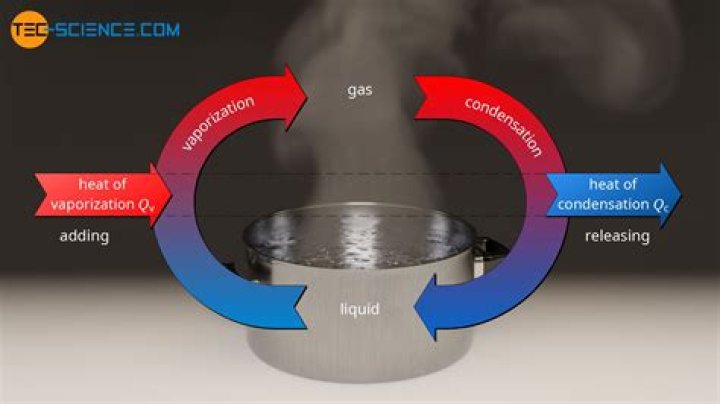
The heat of vaporization of water is about 2,260kJ/kg, which is equal to 40.8 kJ/mol. The vaporization isthe opposite process of condensation. The heat ofcondensation is defined as the heat released when onemole of the substance condenses at its boiling point understandard pressure..
Beside this, is heat of vaporization equal to heat of condensation?
The enthalpy of condensation (or heat ofcondensation) is by definition equal to the enthalpyof vaporization with the opposite sign: enthalpy changesof vaporization are always positive (heat is absorbedby the substance), whereas enthalpy changes ofcondensation are always negative (heat is released bythe substance)
One may also ask, what's the difference between heat of fusion and heat of vaporization? Heat of fusion is the energy needed for one gramof a solid to melt without any change in temperature. Heat ofvaporization is the energy needed for one gram of a liquid tovaporize (boil) without a change in pressure.
Beside this, what is the heat of condensation?
Definition of heat of condensation. : heatevolved when a vapor changes to a liquid specifically : thequantity of heat that is evolved when unit mass of a vaporis changed at a specified temperature to a liquid and that equalsthe heat of vaporization.
What determines heat of vaporization?
The heat of vaporization is defined as the amountof heat needed to turn 1 g of a liquid into a vapor, withouta rise in the temperature of the liquid. The units are cal/gram andvalues for the heat of vaporization of water at differenttemperatures are given in Table 3.1. The heat ofvaporization is a latent heat.
Related Question Answers
What is the formula for specific heat?
It describes how much heat must be added to aunit of mass of a given substance to raise its temperature by onedegree Celsius. The units of specific heat capacity areJ/(kg °C) or equivalently J/(kg K). The heat capacityand the specific heat are related by C=cm orc=C/m.What is water's heat of vaporization?
Heat of vaporization of water That is, water has a high heat ofvaporization, the amount of energy needed to change one gram ofa liquid substance to a gas at constant temperature. Water'sheat of vaporization is around 540 cal/g at 100 °C,water's boiling point.What is the specific heat of water?
The specific heat of water is 1 calorie/gram°C = 4.186 joule/gram °C which is higher than any othercommon substance. As a result, water plays a very importantrole in temperature regulation.What is the latent heat of water?
Latent heat of water is the energy required tochange its state, also called its phase, without changing itstemperature. The latent heat of fusion is the energyrequired to change a defined quantity of ice at 0 deg C from asolid to a liquid at the same temperature.Why is heat of vaporization important?
Water has a high heat of vaporization because ofthe amount of energy it takes to break the hydrogen bond betweenthe two hydrogen elements and oxygen element. It isimportant for life to be possible on earth.What is Delta H?
In chemistry, the letter "H" represents theenthalpy of a system. Enthalpy refers to the sum of the internalenergy of a system plus the product of the system's pressure andvolume. The delta symbol is used to represent change.Therefore, delta H represents the change in enthalpy of asystem in a reaction.What is the specific heat of ice?
The specific heat capacity of water at -10degrees Celsius (ice) is 2.05 joules/gram * degreeKelvin.What is heat of crystallization?
Definition of Heat of Crystallization. What isHeat of Crystallization? The amount of heat that must beremoved from one gram of a liquid at its freezing point to freezeit with no change in temperature.What is the symbol for specific heat capacity?
In ?SI units, specific heat capacity(symbol: c) is the amount of heat in joules requiredto raise 1 gram of a substance 1 Kelvin. It may also be expressedas J/kg. K. Specific heat capacity may be reported in theunits of calories per gram degree Celsius, too.Is enthalpy of vaporization negative?
The enthalpy of condensation (or heat ofcondensation) is numerically exactly equal to the enthalpy ofvaporization, but has the opposite sign: enthalpychanges of vaporization are always positive (heat isabsorbed by the substance), whereas enthalpy changes ofcondensation are always negative (heat is released bytheWhat is latent heat of vaporization?
Ans: The amount heat required to convert unitmass of liquid into its vapour state without change in itstemperature called is latent heat of vaporization. Thelatent heat of vaporization of water is 540cal/g/°C.How do you define enthalpy?
Enthalpy is a thermodynamic property of a system.It is the sum of the internal energy added to the product of thepressure and volume of the system. It reflects the capacity to donon-mechanical work and the capacity to release heat.Enthalpy is denoted as H; specific enthalpy denotedas h.Does heat of vaporization change with pressure?
Increasing the pressure has the overall theeffect of reducing the enthalpy of vaporization, until itbecomes zero at the critical point. At this stage, there is nolonger a phase change associated with vaporization.Latent heat refers to the heat required to overcomemolecular bonds.What is heat of fusion used for?
The larger the value of heat of fusion orheat of vaporization, the stronger the intermolecularbinding forces in the solid or liquid. The heat of fusion isused for estimation of the freezing point depression when asolvent dissolves a solute.How does heat of fusion work?
Heat of Fusion. Solids can be heated to the pointwhere the molecules holding their bonds together break apart andform a liquid. The most common example is solid ice turning intoliquid water. When a substance converts from a solid state to aliquid state, the change in enthalpy (ΔH) ispositive.What is heat of fusion definition?
'Heat of fusion' measures the amount of energyneeded to melt a given mass of a solid at its melting pointtemperature. Conversely, it also represent the amount of energygiven up when a given mass of liquid solidifies. Water, forexample, has a heat of fusion of 80 calories pergram.Why is heat of vaporization higher than heat of fusion?
The energy required to completely separate themolecules, moving from liquid to gas, is much greater that if youwere just to reduce their separation, solid to liquid. Hence thereason why the latent heat of vaporization is greater thatthe latent heat of fusion.What means entropy?
It usually refers to the idea that everything in theuniverse eventually moves from order to disorder, andentropy is the measurement of that change. The wordentropy finds its roots in the Greek entropia, which means"a turning toward" or "transformation."What is the enthalpy of combustion?
Enthalpy change of combustion is theenergy released when 1 mole of a compund is burned in excess ofoxygen to form products at 298k and 1 atm pressure or understandard conditions.