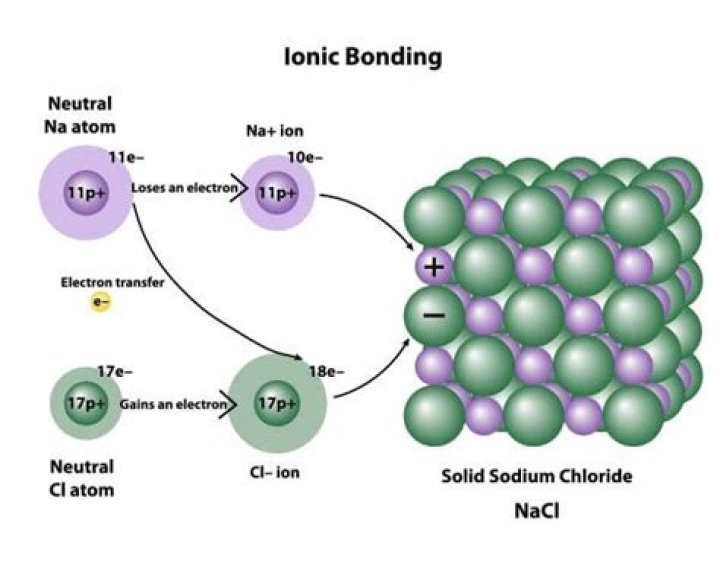
Ionic compounds have high melting and boiling points, so they are in the solid state at room temperature. This energy overcomes the strong electrostatic forces of attraction which act in all directions between the oppositely charged ions: some forces are overcome during melting..
Herein, are all ionic compounds solids at room temperature?
All elemental ionic compounds are solid at room temperature, however there is a class of room temperature ionic liquids. [1] These are a result of poor coordination between the ions in solid form. Typically they involve ions with relatively complex organic components.
Beside above, what are ionic compounds at room temperature? Covalent Bonds vs Ionic Bonds
| Covalent Bonds | Ionic Bonds |
| State at room temperature: | Liquid or gaseous | Solid |
| Polarity: | Low | High |
Similarly, what compound is a solid at room temperature?
An ionic compound is most likely a solid at room temperature and pressure, whereas a covalent compound may be a solid, a liquid, or a gas.
Are all ionic compounds solid?
Ionic compounds consist of oppositely charged ions that are held together by ionic bonds. Ionic compounds are solids with high melting and boiling points. They are good conductors of electricity but only when dissolved in water. Their crystals are rigid and brittle.
Related Question Answers
Will a liquid have ionic bonds?
Can Ionic Compounds Exist as Liquid Under Certain Defined Conditions? Yes. An ionic compound if dissolved in water or melted can exist as liquid as well. Examples include NaCl (salt) dissolved in water, molten electrolytefound in fuel cells.What are 2 properties of ionic substances?
Ionic Compounds have high boiling and melting points as they're very strong and require a lot of energy to break. The electrostatic forces of attraction between oppositely charged ions lead to the formation of ions. Ionic compounds form crystals. These compounds are brittle and break into small pieces easily.Is HCl ionic or covalent?
Sodium chloride is an ionic compound. Many bonds can be covalent in one situation and ionic in another. For instance, hydrogen chloride, HCl, is a gas in which the hydrogen and chlorine are covalently bound, but if HCl is bubbled into water, it ionizes completely to give the H+ and Cl- of a hydrochloric acid solution.How are ionic compounds held together?
An ionic bond is held together by the electrostatic attraction between ions that are near one another. Electrostatic attraction is the attraction between atoms that have opposite charge and holds the atoms together in ionic bonds. Salts, including table salt, are held together by ionic bonds.What are the main properties of ionic compounds?
Properties Shared by Ionic Compounds - They form crystals.
- They have high melting points and high boiling points.
- They have higher enthalpies of fusion and vaporization than molecular compounds.
- They're hard and brittle.
- They conduct electricity when they are dissolved in water.
- They're good insulators.
Which substance is an ionic compound?
Ionic compounds are compounds consisting of ions. Two-element compounds are usually ionic when one element is a metal and the other is a non-metal. Examples include: sodium chloride: NaCl, with Na+ and Cl- ions.Which is responsible for stability of ionic compound?
Charge on the ions. (Magnitude of charge)- higher is the charge, stronger is the electrostatic force of attraction and hence stronger is the ionic bond. Size of the ions- larger the anion and smaller the cation, more is the stability of the compound.Why is salt a solid at room temperature?
Compounds with this kind of electrostatic attraction, called ionic bonds, are almost always solid at room temperature. Above 800°C, however, table salt melts into a liquid, consisting of sodium atoms with positive charges and chloride atoms with negative charges.Are covalent bonds solid at room temperature?
Compounds with covalent bonds may be solid, liquid or gas at room temperature depending on the number of atoms in the compound. Since most covalent compounds contain only a few atoms and the forces between molecules are weak, most covalent compounds have low melting and boiling points.Why ionic compounds are solid and hard?
Ionic compounds are usually hard because they r held together by strong electrostatic forces of attraction. Therefore the oppositely charged ions pack tightly with each other and form a crystalline structure which is very hard to break down.Are metallic bonds solid at room temperature?
chemistry-metallic bonding. Metals have very unique properties. They are strong and usually melt at very high temperatures. Of course the exception to this is mercury which is a liquid at room temperature.How can you tell if a gas is solid or liquid?
When we increase the temperature of a solid it will become liquid at its melting point. At low temperatures than melting point the substance remain as a solid. Then from melting point temperature to boiling point the substance will remain as a liquid. This is the temperature at which a liquid become a gas.What is stronger ionic or covalent bonds?
Covalent is stronger because the 2 atoms involve share 2 or more outer shell electrons. Covalent bonds hold all of your biomolecules together. Ionic bonds are formed when a valence outer shell electron is transferred from one atom to another - a much weaker interaction.What are examples of ionic solids?
However, they do conduct electricity as liquids (or when dissolved in water) since then the ions can move. Examples of ionic solids include NaCl and Ca(CO3). Ionic compounds are all generally found in nature as ionic solids.