Why is aluminum chloride covalent?
.
Likewise, is aluminum chloride ionic or covalent?
Aluminium oxide is an ionic compound, but aluminium chloride is only ionic in the solid state at low temperatures. At higher temperatures it becomes covalent.
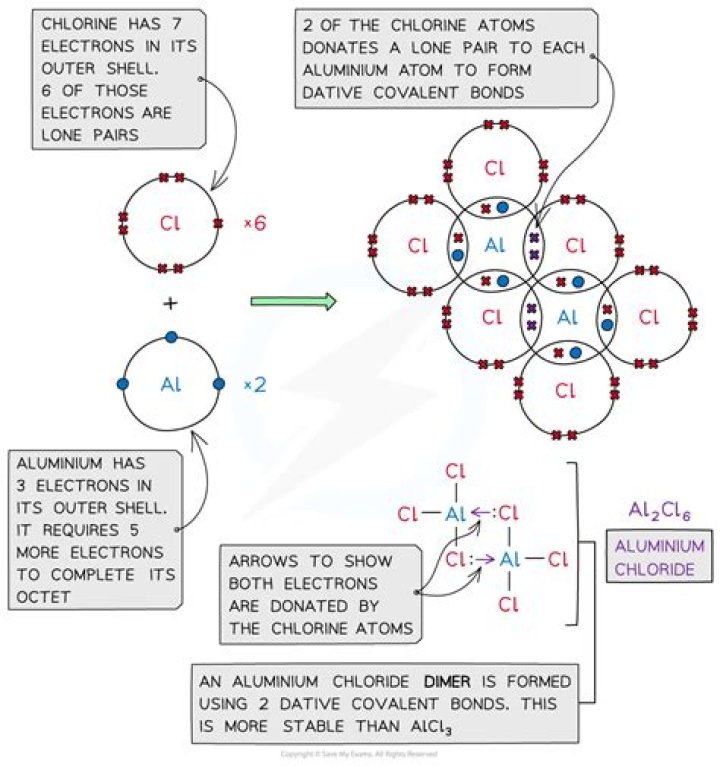
Similarly, why does aluminum chloride exist as a dimer? So, a dative or coordinate bond forms from a Chlorine atom on one molecule to the Aluminium on the other. Consequently, the molecules pair up or dimerize so that each Aluminium atom has a share of eight electrons in its outer shell.
Similarly, you may ask, what type of bond is aluminum chloride?
Explanation: The Al-Cl bond is polar covalent. This is consistent with the fact that aluminum chloride changes directly from a solid to a gas at the relatively low temperature of 180 °C. Al has three valence electrons.
Why is Aluminium chloride AlCl3?
Aluminium Chloride (AlCl3) Uses AlCl3 is used mainly as a catalyst for different chemical reactions. It is used extensively in Friedel-Crafts reaction including both acylations and alkylations. It is used during the preparation of anthraquinone from phosgene and benzene.
Related Question AnswersIs HCl a covalent bond?
Answer and Explanation: HCl, also known as hydrochloric acid, has a covalent bond. The hydrogen (H) atom shares an electron with the chlorine (Cl) to form the bond.Is co2 a covalent bond?
Carbon dioxide is covalent bond. Carbon cannot form ionic bond because in the simplest sense carbon can neither lose 4 electrons for stablisation (Due to it's strong attraction to the nucleus) nor gain 4 electrons (Since it's small nucleus with 6 protons cannot manage extra 4 electrons).Why AlCl3 is covalent but NaCl is ionic?
NaCl is more ionic compared to the ionic nature of AlCl3. In NaCl both cation and anion have low charges while in AlCl3 the cation Al3+, is of a higher charge than Na+ while the anion is the same in both compounds. According to the Fajan's rule, AlCl3 is more covalent in nature.Is magnesium nitride a covalent or ionic bond?
Mg loses two electrons to have an octet. Nitrogen gains three electrons to have an octet. The ionic bond between ions results from the electrostatic attraction of opposite charges. The final formula of magnesium nitride is Mg3N2.Why is AlF3 ionic?
There is this rule called as Fajan's rule which states that with increasing size of the anion and with decreasing size of the cation ionic character of the compound decreases while the covalent character increases. Size of chlorine being greater than fluorine, AlCl3 is covalent while AlF3 is ionic.Why is AlCl3 not ionic?
In case of AlCl3 , though it looks like an ionic reaction between one Aluminium and three Chlorine , it's not that simple. Yes ,it is not. You see , in case of ionic compounds , the metal loses electron while the non-metal accepts the electron. Hence , AlCl3 is basically a covalent compound.Is tap water ionic or molecular?
A molecule of water contains two hydrogen atoms and one oxygen atom which are connected by covalent bonds. Normally, water is a molecular. However, at high temperatures and pressures, it can exist as ionic water, whereby the molecules break into ions of hydrogen and oxygen. Therefore, it can be ionic as well.Is h2o covalent or ionic?
H2O, more commonly known as water, is a covalent compound. This type of compound is the result of atoms, usually from nonmetal elements, sharing electrons. Water has a special type of covalent bond called a polar covalent bond.What type of bond is citric acid?
Citric acid is a covalent, or molecular, compound because it contains single and double covalent bonds formed by the sharing of pairs of electrons between different atoms. The molecular formula of citric acid is C6H8O7.Is nitrogen dioxide a covalent or ionic bond?
Answer and Explanation: NO2 or nitrogen dioxide is a covalent compound. NO2 is comprised of Nitrogen and oxygen which are both highly electronegative chemical elements.What type of bond is CH?
It is what we call a polar covalent bond. When the difference in electronegativity between two atoms is small (say less than about 0.5), like in a C-H bond, then the bond is said to be a non-polar covalent bond, with no descernable polarity.Is AlCl3 polar or nonpolar?
AlCl3 monomer is trigonal planar (similar to BF3), and it is non-polar. The dipole moments of each of the Al-Cl bond is directed at 120 degree angles to each other in a plane, and is therefore cancelled out. Therefore it is a non-polar molecule.Is Al a metal?
Aluminium is usually considered to be a metal, as described in the Wikipedia article Metalloids: Aluminium: Aluminium is ordinarily classified as a metal. It is lustrous, malleable and ductile, and has high electrical and thermal conductivity.Is AlBr3 covalent?
CBr4 and AlBr3 are both not ionic compounds. AlBr3 might seem as though it should be one, but it has a low melting and boiling point and consists of Al2Br6 molecules, not ions. Fajans' rules can be used to estimate whether a compound will be ionic or covalent.Is phosphorus ionic or covalent?
The nonmetal covalent elements found in the periodic table include hydrogen, carbon, nitrogen, phosphorus, oxygen, sulfur and selenium. Additionally, all of the halogen elements, including fluorine, chlorine, bromine, iodine and astatine, are all covalent nonmetal elements.Will aluminum and chlorine form ionic bond?
Because aluminum has three, that means three chlorine atoms can bond. They make the formula AlCl3, also known as aluminum trichloride. Each of the chlorine atoms gets an electron to fill its shell, and the aluminum loses three, giving it a filled shell too (remember, aluminum has three extra electrons).What type of bond is aluminum and fluorine?
It consists of an aluminum atom surrounded by three fluoride atoms, connected with covalent bonds. As aluminum has seven electrons in it's outer shell, while fluoride has three, so those three bond with the aluminum to make a complete shell of eight electrons.How do you make Aluminium chloride?
Synthesis. Aluminium chloride is manufactured on a large scale by the exothermic reaction of aluminium metal with chlorine or hydrogen chloride at temperatures between 650 to 750 °C (1,202 to 1,382 °F). Aluminum chloride may be formed via a single displacement reaction between copper chloride and aluminum metal.What antiperspirants have aluminum chloride?
Popular over the counter antiperspirants for hyperhidrosis| Brand | Forms | Compount |
|---|---|---|
| Driclor | Roll-on, Solution | aluminum chloride. |
| SweatBlock | Wipes | aluminum chloride. |
| Dove | Solid | aluminum zirconium tetrachlorohydrex gly (20%) |
| Secret Clinical Strength | Clear Gel | Aluminum zirconium tetrachlorohydrex Gly |