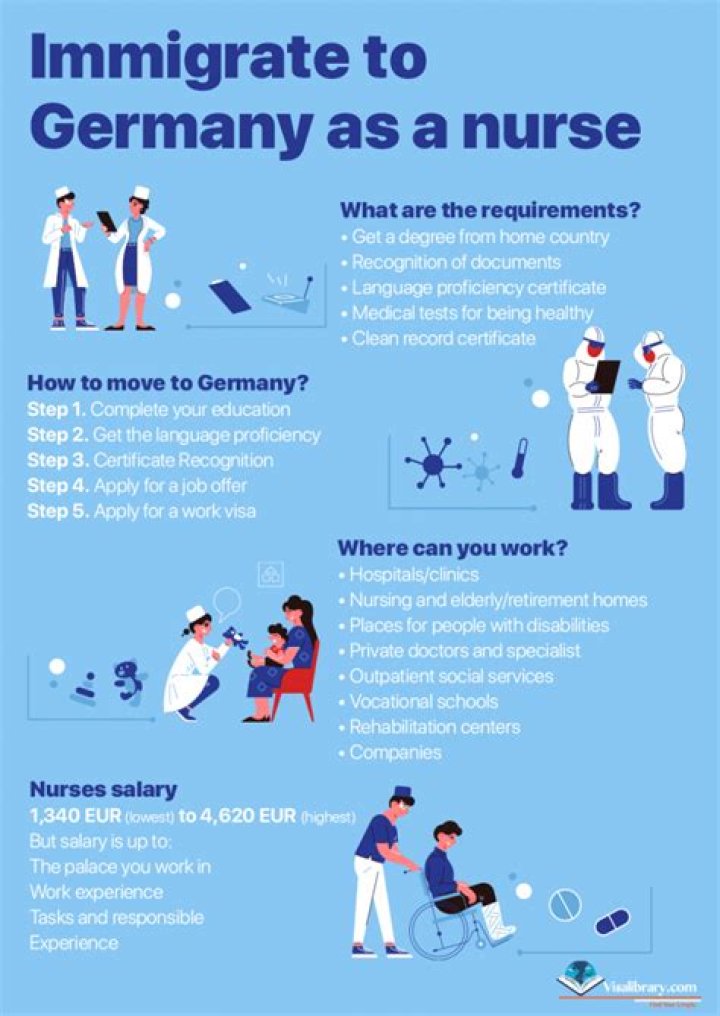
| Name | Magnesium |
| Boiling Point | 1107.0° C |
| Density | 2.62 grams per cubic centimeter |
| Normal Phase | Solid |
| Family | Alkaline Earth Metals |
.
Considering this, why is the melting point of magnesium higher than sodium?
Metallic bonding in magnesium If you work through the same argument with magnesium, you end up with stronger bonds and so a higher melting point. Magnesium atoms also have a slightly smaller radius than sodium atoms, and so the delocalised electrons are closer to the nuclei.
Similarly, does sodium have a higher melting point than magnesium? Use the sea of electrons model to explain why Magnesium has a higher melting point (650 °C) than sodium (97.79 °C). If you work through the same argument above for sodium with magnesium, you end up with stronger bonds and hence a higher melting point. Magnesium has the outer electronic structure 3s2.
Also Know, why does magnesium have a high boiling point?
The higher charge on the Mg2+ ion means there is a stronger electrostatic force of attraction between the ions and the delocalised electrons in the metal lattice, this means that more energy is required to overcome this attraction and so Magnesium has a higher melting point than Sodium.
Why Magnesium has lower melting point?
The melting points get lower as you go down the Group because the metallic bonds get weaker. The oddity of magnesium has to be explained separately. The atoms in a metal are held together by the attraction of the nuclei to the delocalised electrons.
Related Question Answers
Is sodium stronger than magnesium?
Sodium metal is therefore written as Na - not Na+. If you work through the same argument with magnesium, you end up with stronger bonds and so a higher melting point. Magnesium atoms also have a slightly smaller radius than sodium atoms, and so the delocalised electrons are closer to the nuclei.Does magnesium conduct electricity?
Sodium, magnesium and aluminium are all metals. They have metallic bonding, in which the nuclei of metal atoms are attracted to delocalised electrons. there are more electrons that can move and carry charge through the structure … the electrical conductivity increases.Why is magnesium malleable?
Its because the atoms of Mg hold themselves in a regular lattice structured grouping, meaning in a regular order of arrangement. So when you hit Mg on one side, its layer slide to the direction of the force changes its shape without molecular change therefore Mg is considered malleable.What is the flash point of magnesium?
Magnesium is also flammable, burning at a temperature of approximately 2500 K (2200 °C, 4000 °F). The autoignition temperature of magnesium is approximately 744 K (473 °C, 883 °F).Why is the melting point of sodium chloride high?
Sodium chloride has a high melting point because the lattice is constructed from positive sodium ions and negative chloride ions. This ions attract each other with a strong electrostatic force. This leads to a strong construction. In salts with ion charge 2+ and 2- the melting point is in general much higher.Why do diamond and graphite have such high melting points?
Both diamond and graphite have a giant molecular structure. The carbon atoms are held together by strong covalent bonds which require high temperatures to break. This causes diamond and graphite to have high melting points.Why is the melting point of Aluminium lower than magnesium?
Melting and boiling points rise across the three metals because of the increasing strength of the metallic bonds. The number of electrons which each atom can contribute to the delocalised "sea of electrons" increases. The atoms also get smaller and have more protons as you go from sodium to magnesium to aluminium.Does magnesium conduct heat?
A characteristic of metals that makes them good conductors is that they have mobile valence electrons. However besides that, magnesium is a bad metal to make electrical contacts and wires from. Pure magnesium forms a hard, thin oxide on the surface on contact with air. The oxide is very low conductivity.Is magnesium malleable?
Magnesium is a ductile, silver-white, chemically active metal with a hexagonal close-packed crystalline structure. It is malleable when heated. When heated, magnesium powder or ribbon ignites and burns with an intense white light and releases large amounts of heat, forming the oxide, magnesia , MgO.How do you extinguish a magnesium fire?
Burning magnesium is usually quenched by using a Class D dry chemical fire extinguisher, or by covering the fire with sand to remove its air source.What is the heaviest element in Group 2?
Radium (Ra) Radium is the heaviest and most radioactive of the alkaline earth metals and it reacts explosively with water.Why do transition metals have high melting points?
The melting-points of the transition metals are high due to the 3d electrons being available for metallic bonding. The densities of the transition metals are high for the same reason as the high boiling points. These properties are the result of metallic bonding between the atoms in the metal lattice.Why are metals soft and malleable?
Metals are described as malleable (can be beaten into sheets) and ductile (can be pulled out into wires). This is because of the ability of the atoms to roll over each other into new positions without breaking the metallic bond.Which metal has the strongest metallic bonding?
Molybdenum
Why beryllium has high melting point?
As this ion is so small it is highly polarising, to the extent that its compounds are rather covalent. When it forms a metal it means that the bonds are very high energy relative to the atomic size which causes the atoms to be bound tightly. This means its requires more energy (higher temperature) to break the bonds.Why is the melting point of sodium lower than Aluminium?
Melting point increases for metals Na, Mg and Al Therefore metallic bond becomes stronger and melting point increases from sodium to aluminium. In fact the metallic bond in sodium and other Group 1 metals is so weak that the melting point of sodium is unusually low and falls within the region of simple molecules.What are Group 2 elements called?
The alkaline earth metals are six chemical elements in group 2 of the periodic table. They are beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra).Why are metallic bonds so strong?
The electrons and the positive ions in the metal have a strong attractive force between them. Metallic bonds causes many of the traits of metals, such as strength, malleability, ductility, luster, conduction of heat and electricity. Because the electrons move freely, the metal has some electrical conductivity.What is potassium's freezing point?
Freezing point of Potassium (K) is 63.38 °C [ Convert 63.38 °C to different units ]