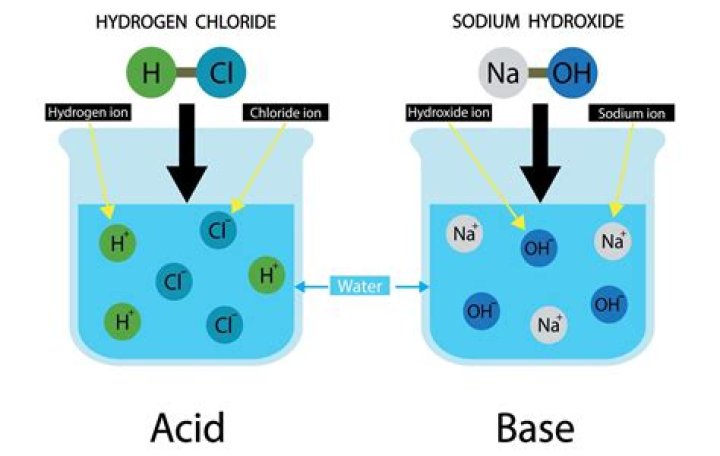
What produces H+ ions in a solution?
.
Likewise, which produces H+ ions?
Acids
Secondly, why do basic solutions have H+ ions? The reason is that water on its own splits into hydrogen and hydroxide ions. When a solution is very basic, the extra hydroxide ions will react with any hydronium almost as soon as it forms. Thus the concentration of hydrogen ions is very low, so low that it can be usually ignored.
Thereof, what is a substance that produces H+ ions in a water solution?
Acids, Bases and Solutions - Mrs. Henderson
| A | B |
|---|---|
| Any substance that produces hydrogen ions in water | acid |
| What is the hydroxide ion? | a negative ion, made of oxygen and hydrogen |
| any substance that produces hydroxide ions in water | base |
| How cn you tell the concentration of hydrogen ions in a solution? | pH scale |
What type of solution is formed when the H+ ions equal the OH ions?
A neutral solution will have H+ ions equal to OH- ions. A neutral substance will have a pH of 7. > An example of a neutral solution is water: the # of H+ ions = # of OH- ions. > If water is combined with more H+ ions, it will become more acidic. >
Related Question AnswersWhich is more stable H+ or H?
Originally Answered: Why is H+ ion more stable as compared to H- ion? Let's first think about it this way. A H+ ion will contain no electrons while a H- electron will contain more than 1 electrons. This is because the proton in the hydrogen atom can only stabilize 1 electron.Is H+ an acid or base?
But yes, theoretically speaking H+ is a Brønsted–Lowry acid as the definition of such is to donate a proton and it donates itself to water in the case. It's also a Lewis acid as by donating itself it accepts electrons in its 1s emptry orbital.What is the name for the H+ ions?
Hydrogen ion, strictly, the nucleus of a hydrogen atom separated from its accompanying electron. The hydrogen nucleus is made up of a particle carrying a unit positive electric charge, called a proton (q.v.). The isolated hydrogen ion, represented by the symbol H+, is therefore customarily used to represent a proton.Is hydroxyl an ion?
Hydroxide is a diatomic anion with chemical formula OH−. The corresponding covalently-bound group –OH of atoms is the hydroxy group. Hydroxide ion and hydroxy group are nucleophiles and can act as a catalysts in organic chemistry.Why is H+ a proton?
Why is a H+ ion referred to as a proton? An atom of hydrogen contains 1 proton, 1 electron and 0 neutrons. When hydrogen loses an electron to become H+ only a proton remains. We can work out the number of neutrons an atom has by deducting the atomic number from the mass number.Is HCl a Bronsted acid?
The Brønsted-Lowry Theory of Acids and Bases Therefore, HCl is a Brønsted-Lowry acid (donates a proton) while the ammonia is a Brønsted-Lowry base (accepts a proton). Also, Cl- is called the conjugate base of the acid HCl and NH4+ is called the conjugate acid of the base NH3.Which acid is stronger hydrochloric or acetic?
HCl liberates more H+ ions while in contacts with water as compared to acetic acid so HCl is a strong acid but Acetic acid is a weak acid. Even the concentration of hydrogen ions lowers the pH. Generally, Hydrochloric acid is considered as a strong acid and ethanoic acid is a weak acid.Is H+ A gas?
H+ can exist as such only in an ionized gas phase (and, perhaps, in metals such as palladium where hydrogen was dissolved). In liquid solutions, it immediately bonds to the strongest base there available, since it is a very strong Lewis acid. So, in water, it becomes immediately H3O+ ion.Is soapy water neutral?
Tap water is usually a 7 (neutral) while soapy water is influenced by the alkaline in the soap and measures a 12.What is a substance that produces OH ions in a solution?
base. a substance that produces OH- ions in solution. hydronium ion.Does ch4 produce H+ ions?
CH4 has a neutral pH, around 7. On the other hand, substances such as hydrochloric acid, HCl, are held together by polar ionic bonds and when placed into water the hydrogen will break away to form hydrogen ions, making the liquid acidic.What unit are known as hydronium ions?
What is the concentration of hydronium ions (H3O+) or free unbound protons (H+) as an equivalent of pH=7.4 in water solution? From definition of pH is known that an activity of hydrogen ions is 10^(-pH). The activity of substance is activity coeficient multiplied by physical concentration (in units "mol/mol").What produces hydroxide ions in water?
In this reaction, sodium hydroxide (NaOH) disassociates into sodium (Na+) and hydroxide (OH-) ions when dissolved in water, thereby releasing OH- ions into solution. Arrhenius acids are substances which produce hydrogen ions in solution. Arrhenius bases are substances which produce hydroxide ions in solution.What has the highest pH?
By definition, the strongest base in water is hydroxide (OH-). Any compound that dissociates completely and is a hydroxide can produce the highest pH (i.e., 14).When an acid is dissolved in water H+ ions form?
An Arrhenius acid is any substance that ionizes when it dissolves in water to give the H+, or hydrogen, ion. An Arrhenius base is any substance that gives the OH-, or hydroxide, ion when it dissolves in water.Does ch3cooh produce H+ ions?
CH3COOH is a weak acid and dissociates partially in solution (as indicated with reversible arrow) to form H+ and CH3COO- ions.How do acids feel?
Acids taste sour while bases taste bitter. An acid reacts with metals to produce bubbles of hydrogen gas while a base feels slimy to the touch.What has a pH of 9?
What does it mean for a solution to be acidic or basic (alkaline)?| pH Value | H+ Concentration Relative to Pure Water | Example |
|---|---|---|
| 8 | 0.1 | sea water, eggs |
| 9 | 0.01 | baking soda |
| 10 | 0.001 | Great Salt Lake, milk of magnesia |
| 11 | 0.000 1 | ammonia solution |